Keywords
1-(2-hydrazinoaryl)-3,4-dihydroisoquinolines
7-azaindazoles
b-phenylethylamines
indazoles
isoquinolines
recyclization
Abstract
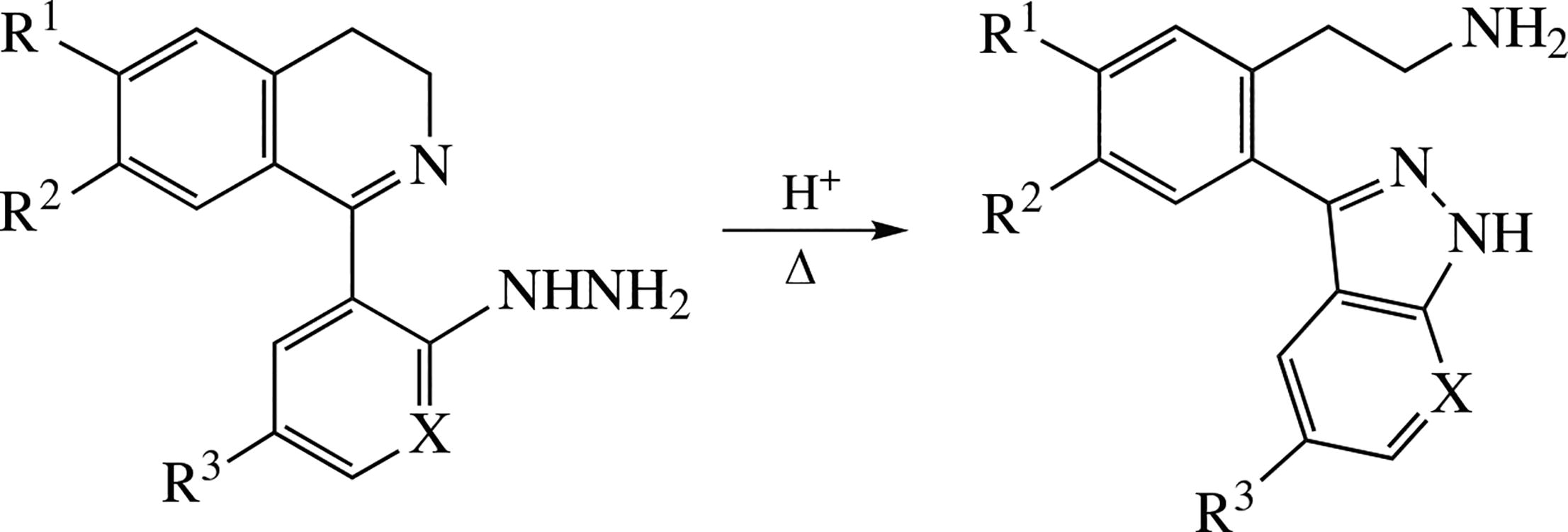
1-(2-Hydrazinoaryl)-3,4-dihydroisoquinolines and their hydrazinopyridyl analogues undergo recyclization affording novel (7-aza)indazolyl-β-arylethylamines. The products are expected to possess neurotropic activities.
References
1.

Qin J., Cheng W., Duan Y., Yang H., Yao Y.
Anti-Cancer Agents in Medicinal Chemistry,
2020
2.

Panda N., Karmakar S., Jena A.K.
Chemistry of Heterocyclic Compounds,
2011
3.

Le Manach C., Paquet T., Brunschwig C., Njoroge M., Han Z., Gonzàlez Cabrera D., Bashyam S., Dhinakaran R., Taylor D., Reader J., Botha M., Churchyard A., Lauterbach S., Coetzer T.L., Birkholtz L., et. al.
Journal of Medicinal Chemistry,
2015
4.

5.

Manjunatha U.H., Vinayak S., Zambriski J.A., Chao A.T., Sy T., Noble C.G., Bonamy G.M., Kondreddi R.R., Zou B., Gedeck P., Brooks C.F., Herbert G.T., Sateriale A., Tandel J., Noh S., et. al.
Nature,
2017
6.

Rice K.D., Kim M.H., Bussenius J., Anand N.K., Blazey C.M., Bowles O.J., Canne-Bannen L., Chan D.S., Chen B., Co E.W., Costanzo S., DeFina S.C., Dubenko L., Engst S., Franzini M., et. al.
Bioorganic and Medicinal Chemistry Letters,
2012
7.

Orlikova B., Chaouni W., Schumacher M., Aadil M., Diederich M., Kirsch G.
European Journal of Medicinal Chemistry,
2014
8.

El-Borai M.A., Rizk H.F., Beltagy D.M., El-Deeb I.Y.
European Journal of Medicinal Chemistry,
2013
9.

Bare T.M., McLaren C.D., Campbell J.B., Firor J.W., Resch J.F., Walters C.P., Salama A.I., Meiners B.A., Patel J.B.
Journal of Medicinal Chemistry,
1989
10.

Saito M.S., Lourenço A.L., Dias L.R., Freitas A.C., Vitorino M.I., Albuquerque M.G., Rodrigues C.R., Cabral L.M., Dias E.P., Castro H.C., Satlher P.C.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2016
11.

Blass B.
ACS Medicinal Chemistry Letters,
2015
12.

Hamblin J.N., Angell T.D., Ballantine S.P., Cook C.M., Cooper A.W., Dawson J., Delves C.J., Jones P.S., Lindvall M., Lucas F.S., Mitchell C.J., Neu M.Y., Ranshaw L.E., Solanke Y.E., Somers D.O., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
13.

Beatty J.W., Lindsey E.A., Thomas-Tran R., Debien L., Mandal D., Jeffrey J.L., Tran A.T., Fournier J., Jacob S.D., Yan X., Drew S.L., Ginn E., Chen A., Pham A.T., Zhao S., et. al.
Journal of Medicinal Chemistry,
2020
14.
DrugBank, https://www.drugbank.com.
15.

Deibler K.K., Schiltz G.E., Clutter M.R., Mishra R.K., Vagadia P.P., O'Connor M., George M.D., Gordon R., Fowler G., Bergan R., Scheidt K.A.
ChemMedChem,
2019
16.
Protopopov M.V., Vdovin V.S., Lukashov S.S., Ostrynska O.V., Borysenko I.P., Borovykov O.V., Starosyla S.A., Bilokin Y.V., Kukharenko O.P., Bdzhola V.G., Yarmoluk S.M.
Biopolymers and Cell,
2020
17.

Chattha F.A., Kousar S., Mehr-un-Nisa, Munawar M.A.
Letters in Drug Design and Discovery,
2018
18.

Rai G.S., Maru J.J.
Chemistry of Heterocyclic Compounds,
2020
19.
Trace Amines and Neurological Disorders: Potential Mechanisms and Risk Factors, eds. T. Farooqui and A. A. Farooqui, Elsevier, 2016.
20.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Kartsev V.G., Sochnev V.S., Klimenko A.I., Dobaeva N.M., Borodkin G.S., Bodryakov A.N., Bodryakova M.A., Fetisov L.N.
Mendeleev Communications,
2021
21.
A. A. Zubenko, A. S. Morkovnik, L. N. Divaeva, V. S. Sochnev, O. P. Demidov, A. N. Bodryakov, L. N. Fetisov, K. N. Kononenko, M. A. Bodryakova and A. I. Klimenko, Russ. J. Gen. Chem., 2021, 91, (Zh. Obshch. Khim., 2021, 91, 703).
22.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Sochnev V.S., Borodkina I.G., Drobin Y.D., Spasov A.A.
Mendeleev Communications,
2020
23.

Gaikwad D.D., Chapolikar A.D., Devkate C.G., Warad K.D., Tayade A.P., Pawar R.P., Domb A.J.
European Journal of Medicinal Chemistry,
2015
24.

Palumbo Piccionello A., Pace A., Pibiri I., Buscemi S., Vivona N.
Tetrahedron,
2006
25.

Palumbo Piccionello A., Pace A., Pierro P., Pibiri I., Buscemi S., Vivona N.
Tetrahedron,
2009
26.

Gallardo-Fuentes S., Contreras R.
Organic and Biomolecular Chemistry,
2015
27.

Akopova A.R., Morkovnik A.S., Khrustalev V.N., Bicherov A.V.
Russian Chemical Bulletin,
2013
28.

Tatto M.T.
Oxford Review of Education,
2015
29.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
30.
G. M. Sheldrick, Acta Crystallogr., 2015, C71, 3.
31.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009