Keywords
N-vinylpyridinium salts
acetylenedicarboxylates
cascade reactions
cycloheptatrieneheptacarboxylates
diastereomers
Abstract
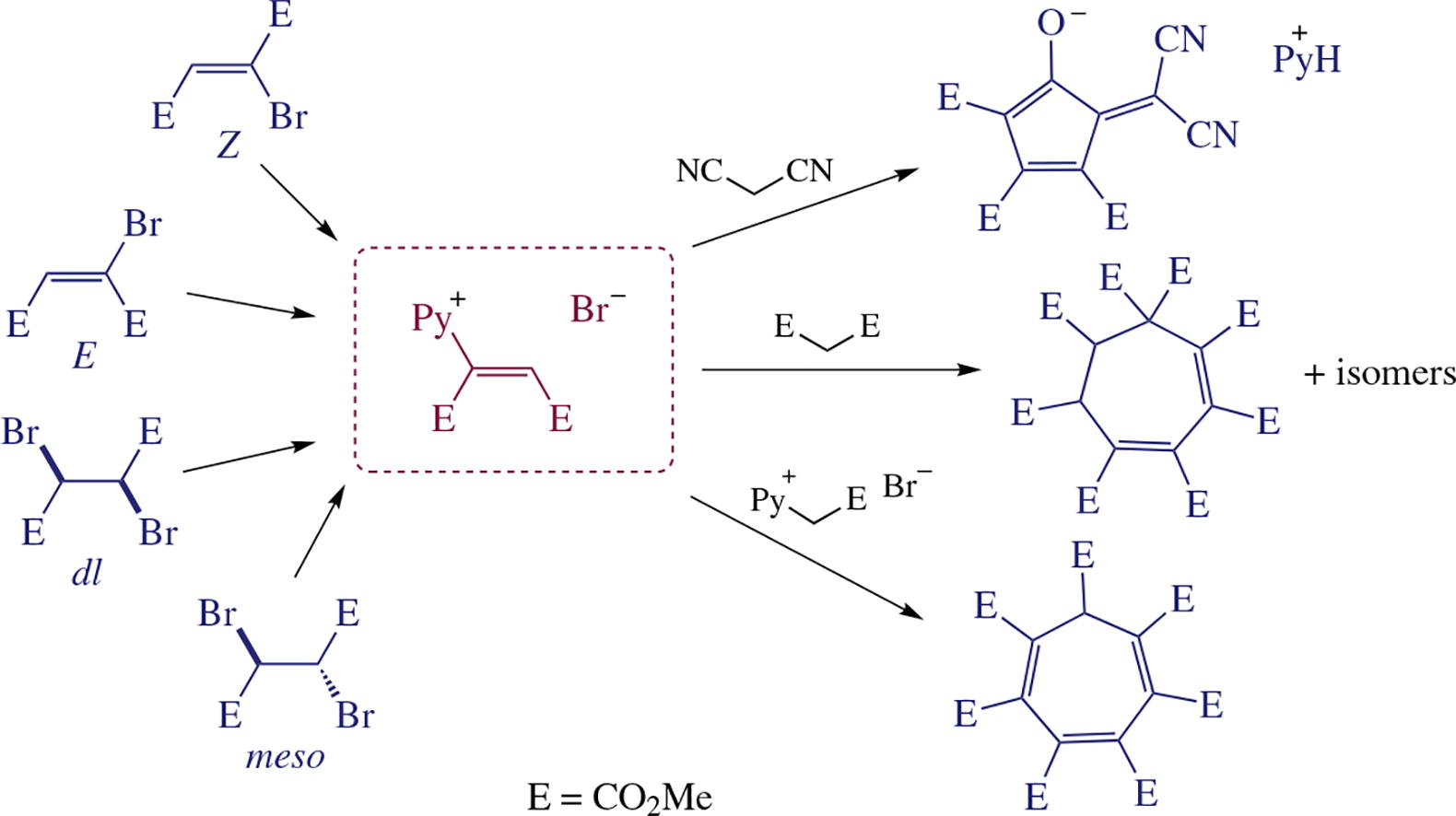
The configuration of N-[1,2-bis(methoxycarbonyl)vinyl]-pyridinium intermediate generated from stereoisomers of dimethyl dibromosuccinate or dimethyl bromobutenedioate does not depend on the precursors’ configuration, as distinct from the yields and the reaction rates. The use of various nucleophiles in these cascade reactions gives either cycloheptadieneoctacarboxylic or cycloheptatrienehepta-carboxylic esters along with pyridinium 5-dicyano-methylidene-2,3,4-tris(methoxycarbonyl)cyclopenta-1,3-dien-1-olate. In the latter case, the acyclic ‘Diels’ golden adduct’ was detected, and its structure was refined.
References
1.

Mikhailov I.E., Dushenko G.A., Minkin V.I.
Russian Journal of Organic Chemistry,
2021
2.

Grondal C., Jeanty M., Enders D.
Nature Chemistry,
2010
3.

Pellissier H.
Organic Preparations and Procedures International,
2019
4.
Tukhtaev H.B., Sorokin I.D., Melnikov M.Y., Budynina E.M.
Mendeleev Communications,
2020
5.
Schmidt E.Y., Semenova N.V., Ivanova E.V., Bidusenko I.A., Trofimov B.A.
Mendeleev Communications,
2020
6.
Zyryanov G.V., Kopchuk D.S., Kovalev I.S., Santra S., Rahman M., Khasanov A.F., Krinochkin A.P., Taniya O.S., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
7.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Leonova N.A., Egorov M.P.
Mendeleev Communications,
2020
8.

Stephanidou-Stephanatou J., Neochoritis C., Zarganes-Tzitzikas T.
Synthesis,
2014
9.

Shaabani A., Bazgir A., Tavasoli-Rad F., Bijanzadeh H.R., Razmara F.
Journal of Chemical Research,
2004
10.

Shaabani A., Rezayan A.H., Sarvary A., Heidary M., Ng S.W.
Tetrahedron,
2009
11.

Shaabani A., Soleimani E.
Phosphorus, Sulfur and Silicon and the Related Elements,
2006
12.

Xia E., Sun J., Yao R., Yan C.
Tetrahedron,
2010
13.

Yavari I., Anary-Abbasinejad M., Alizadeh A.
Monatshefte fur Chemie,
2002
14.
15.

Huisgen R., Morikawa M., Herbig K., Brunn E.
Chemische Berichte,
1967
16.

Diels O.
Berichte der deutschen chemischen Gesellschaft (A and B Series),
1942
17.

Goff E.L., LaCount R.B.
Journal of Organic Chemistry,
1964
18.

Salikov R.F., Trainov K.P., Platonov D.N., Belyy A.Y., Tomilov Y.V.
European Journal of Organic Chemistry,
2018
19.

Belyy A., Platonov D., Salikov R., Levina A., Tomilov Y.
Synlett,
2018
20.

Trainov K.P., Chechekina O.G., Salikov R.F., Platonov D.N., Tomilov Y.V.
Dyes and Pigments,
2021
21.

Development of a platform for the discovery and practical synthesis of new tetracycline antibiotics.
Liu F., Myers A.G.
Current Opinion in Chemical Biology,
2016
22.

Rennison D., Bova S., Cavalli M., Ricchelli F., Zulian A., Hopkins B., Brimble M.A.
Bioorganic and Medicinal Chemistry,
2007
23.

Kendall J.T., Lemal D.M.
Journal of Fluorine Chemistry,
1997
24.

Hudlicky M.
Journal of Fluorine Chemistry,
1972
25.

Salikov R.F., Trainov K.P., Platonov D.N., Davydov D.A., Lee S., Gerasimov I.S., Medvedev M.G., Levina A.A., Belyy A.Y., Tomilov Y.V.
Dyes and Pigments,
2019
26.

Belyy A.Y., Platonov D.N., Salikov R.F., Trainov K.P., Medvedev M.G., Luponosov Y.N., Svidchenko E.A., Tomilov Y.V.
Dyes and Pigments,
2021
27.
10.1016/j.mencom.2022.03.037_b0135
Cookson
J. Chem. Soc., C,
1986
28.
10.1016/j.mencom.2022.03.037_b0140
Erickson
J. Am. Chem. Soc.,
1867