Keywords
$\alpha$-amino phosphonates
$\gamma$,$\delta$-didehydro lysine
hydrogenation
Mannich reaction
α-amino carboxylates
Abstract
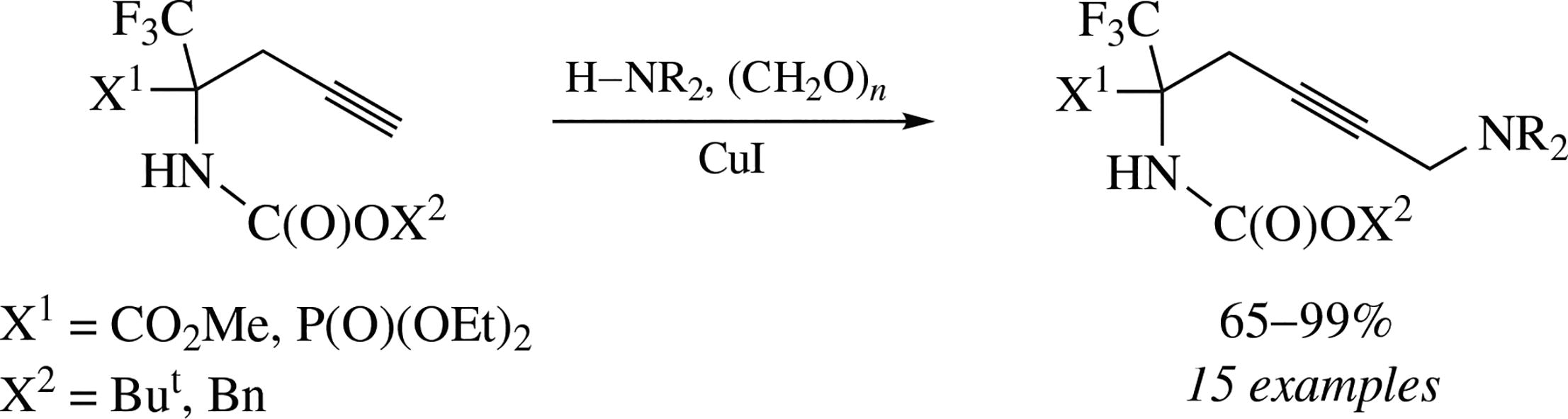
An efficient and selective access to novel α-CF3-substituted γ,δ-didehydro lysine derivatives and their phosphorus analogues has been developed via the Cu-catalyzed Mannich reaction of α-amino α-propargyl α-trifluoromethyl carboxylates or phosphonates with different secondary amines and paraformaldehyde.
References
1.
(a) F. P. J. T. Rutjes, L. B. Wolf and H. E. Schoemaker, J. Chem. Soc., Perkin Trans. 1, 2000, 4197
2.

Jin S., Xu M.
Advanced Synthesis and Catalysis,
2010
3.

Masuda Y., Maruyama C., Kawabata K., Hamano Y., Doi T.
Tetrahedron,
2016
4.
4-(3-fluoropropyl) arginine as a tumor imaging agent](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wu R., Liu S., Liu Y., Sun Y., Cheng X., Huang Y., Yang Z., Wu Z.
European Journal of Medicinal Chemistry,
2019
5.

Kaiser J., Kinderman S.S., van Esseveldt B.C., van Delft F.L., Schoemaker H.E., Blaauw R.H., Rutjes F.P.
Organic and Biomolecular Chemistry,
2005
6.

Jones M.A., Hislop A.D., Snaith J.S.
Organic and Biomolecular Chemistry,
2006
7.

Peng L., He Z., Xu X., Guo C.
Angewandte Chemie - International Edition,
2020
8.

Al Temimi A.H., White P.B., Mulders M.J., van der Linden N.G., Blaauw R.H., Wegert A., Rutjes F.P., Mecinović J.
Chemical Communications,
2020
9.

de Bruin G., van Rooden E.J., Ward D., Wesseling C., van den Nieuwendijk A.M., van Boeckel C.A., Driessen C., Kisselev A.F., Florea B.I., van der Stelt M., Overkleeft H.S.
European Journal of Organic Chemistry,
2017
10.

Smits R., Cadicamo C.D., Burger K., Koksch B.
Chemical Society Reviews,
2008
11.

Moschner J., Stulberg V., Fernandes R., Huhmann S., Leppkes J., Koksch B.
Chemical Reviews,
2019
12.

Bacchi C.J., Goldberg B., Garofalo-Hannan J., Rattendi D., Lyte P., Yarlett N.
Biochemical Journal,
1995
13.

Perrin S.R., Hauck W., Ndzie E., Blehaut J., Ludemann-Hombouger O., Nicoud R., Pirkle W.H.
Organic Process Research and Development,
2007
14.

Raul F.
Biochemical Society Transactions,
2007
15.

Köckinger M., Ciaglia T., Bersier M., Hanselmann P., Gutmann B., Kappe C.O.
Green Chemistry,
2018
16.

Philippova A.N., Vorobyeva D.V., Monnier F., Osipov S.N.
Organic and Biomolecular Chemistry,
2020
17.

Shchetnikov G.T., Zotova M.A., Bruneau C., Dixneuf P.H., Osipov S.N.
European Journal of Organic Chemistry,
2010
18.
![Synthesis of functionalized CF3-containing heterocycles via [2,3]-sigmatropic rearrangement and sequential catalytic carbocyclization](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Vorobyeva D.V., Mailyan A.K., Peregudov A.S., Karimova N.M., Vasilyeva T.P., Bushmarinov I.S., Bruneau C., Dixneuf P.H., Osipov S.N.
Tetrahedron,
2011
19.
![Thermal [2+2] Cycloaddition of CF3-Substituted Allenynes: Access to Novel Cyclobutene-Containing α-Amino Acids](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Osipov S., Mailyan A., Krylov I., Bruneau C., Dixneuf P.
Synlett,
2011
20.
(c) A. K. Mailyan, A. S. Peregudov, P. H. Dixneuf, C. Bruneau and S. N. Osipov, J. Org. Chem., 2012, 77, 8518.
21.

Rona P., Crabbe P.
Journal of the American Chemical Society,
1969
22.

Crabbé P., Fillion H., André D., Luche J.
Journal of the Chemical Society Chemical Communications,
1979
23.

Convenient methods for the synthesis of highly functionalized and naturally occurring chiral allenes
Periasamy M., Reddy P.O., Sanjeevakumar N.
Tetrahedron Asymmetry,
2014
24.

Jansen A.C., Weustink R.J., Kerling K.E., Havinga E.
Recueil des Travaux Chimiques des Pays-Bas,
2010
25.

Bieber L.W., da Silva M.F.
Tetrahedron Letters,
2004
26.

Romanenko V.D., Kukhar V.P.
Chemical Reviews,
2006
27.

Ordóñez M., Sayago F.J., Cativiela C.
Tetrahedron,
2012
28.

Naydenova E.D., Todorov P.T., Troev K.D.
Amino Acids,
2009
29.

Orsini F., Sello G., Sisti M.
Current Medicinal Chemistry,
2009
30.

Zhao D., Wang R.
Chemical Society Reviews,
2012
31.

Vorobyeva D.V., Karimova N.M., Vasilyeva T.P., Osipov S.N., Shchetnikov G.T., Odinets I.L., Röschenthaler G.
Journal of Fluorine Chemistry,
2010