Keywords
aryl bromides
cross-coupling
ethers
Pd-catalysis
primary alcohols
solvent-free reactions
Abstract
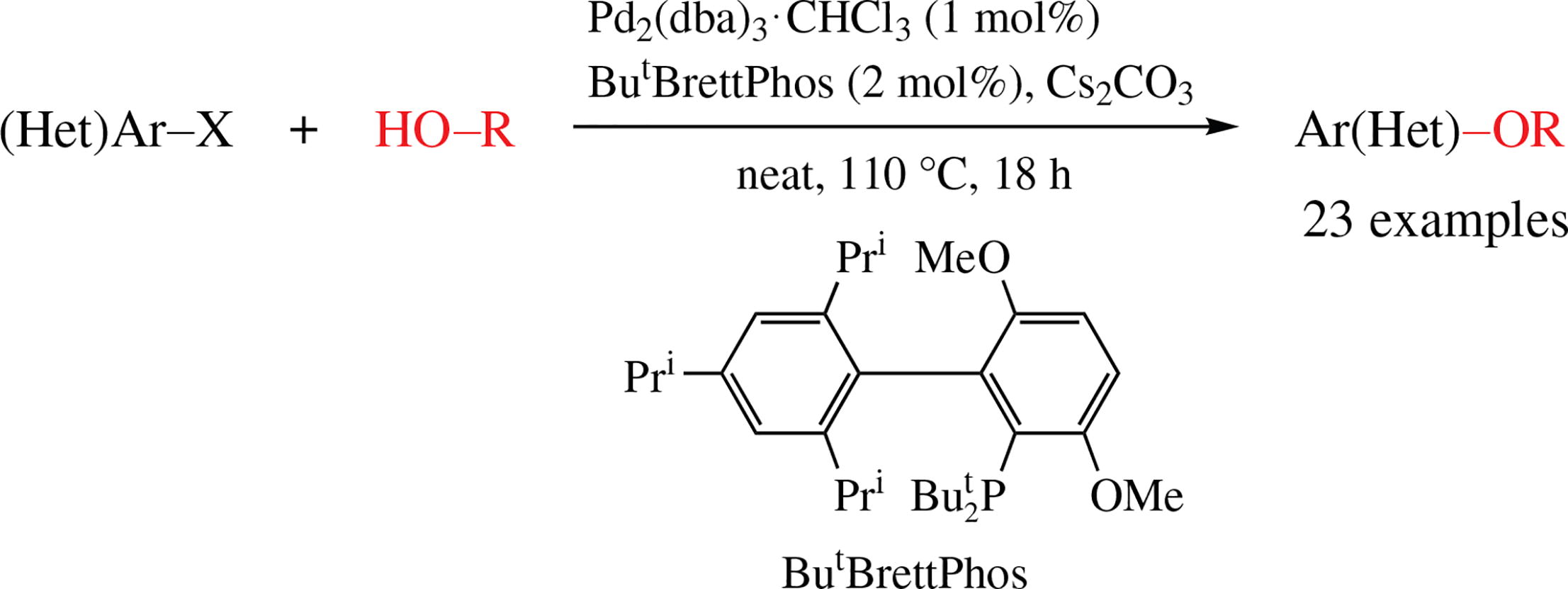
A new efficient solvent-free procedure for the C–O cross-coupling between (hetero)aryl halides with phenols, primary alkanols, or (hetero)arylmethanols using Pd2(dba)3/ButBrettPhos catalytic system is proposed using 23 examples.
References
1.
Varakutin A.E., Muravsky E.A., Shinkarev I.Y., Khrustalev V.N., Semenov V.V.
Mendeleev Communications,
2020
2.
Vereshchagin A.N., Frolov N.A., Konyuhova V.Y., Dorofeeva E.O., Hansford K.A., Egorov M.P.
Mendeleev Communications,
2020
3.
Zubenko A.D., Egorova B.V., Zamurueva L.S., Kalmykov S.N., Fedorova O.A.
Mendeleev Communications,
2021
4.
Le A.T., Tran V.T., Truong H.H., Nguyen L.M., Luong D.M., Do T.T., Nguyen D.T., Dao N.T., Le D.T., Soldatenkov A.T., Khrustalev V.N.
Mendeleev Communications,
2019
5.

Enthaler S., Company A.
Chemical Society Reviews,
2011
6.

Ley S.V., Thomas A.W.
Angewandte Chemie - International Edition,
2003
7.

Cristau P., Vors J., Zhu J.
Tetrahedron,
2003
8.

Theil F.
Angewandte Chemie - International Edition,
1999
9.

Roughley S.D., Jordan A.M.
Journal of Medicinal Chemistry,
2011
10.

Monastyrskyi A., Brockmeyer F., LaCrue A.N., Zhao Y., Maher S.P., Maignan J.R., Padin-Irizarry V., Sakhno Y.I., Parvatkar P.T., Asakawa A.H., Huang L., Casandra D., Mashkouri S., Kyle D.E., Manetsch R., et. al.
Journal of Medicinal Chemistry,
2021
11.

Chen T., Xiong H., Yang J., Zhu X., Qu R., Yang G.
Journal of Agricultural and Food Chemistry,
2020
12.

Ray R., Hartwig J.F.
Angewandte Chemie - International Edition,
2021
13.

Cai Q., Zhou W.
Chinese Journal of Chemistry,
2020
14.

Sambiagio C., Marsden S.P., Blacker A.J., McGowan P.C.
Chemical Society Reviews,
2014
15.

Beletskaya I.P., Cheprakov A.V.
Coordination Chemistry Reviews,
2004
16.

Ullmann F.
Berichte der deutschen chemischen Gesellschaft,
1904
17.

18.

Minnick J.L., Domyati D., Ammons R., Tahsini L.
Frontiers in Chemistry,
2019
19.
(a) Z.W. Williamson, in: Z. Wang (Ed.), Comprehensive Organic Name Reactions and Reagents, John Wiley & Sons, Hoboken, NJ, 2010, pp. 3026−3030
20.

Fuhrmann E., Talbiersky J.
Organic Process Research and Development,
2005
21.

Swamy K.C., Kumar N.N., Balaraman E., Kumar K.V.
Chemical Reviews,
2009
22.
10.1016/j.mencom.2022.03.035_b0025
Caron
Practical Synthetic Organic Chemistry: Reactions, Principles, and Techniques,
2011
23.

Matsushita N., Kashihara M., Formica M., Nakao Y.
Organometallics,
2021
24.

Gowrisankar S., Neumann H., Beller M.
Chemistry - A European Journal,
2012
25.

Milton E.J., Fuentes J.A., Clarke M.L.
Organic and Biomolecular Chemistry,
2009
26.

Rangarajan T.M., Brahma R., Ayushee, Prasad A.K., Verma A.K., Singh R.P.
Tetrahedron Letters,
2015
27.

Wu X., Fors B.P., Buchwald S.L.
Angewandte Chemie - International Edition,
2011
28.

Cheung C.W., Buchwald S.L.
Organic Letters,
2013
29.

Zhang H., Ruiz-Castillo P., Buchwald S.L.
Organic Letters,
2018
30.

Maligres P.E., Li J., Krska S.W., Schreier J.D., Raheem I.T.
Angewandte Chemie - International Edition,
2012
31.

Widenhoefer R.A., Buchwald S.L.
Journal of the American Chemical Society,
1998
32.

Sawatzky R.S., Hargreaves B.K., Stradiotto M.
European Journal of Organic Chemistry,
2016
33.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019
34.

Rzhevskiy S.A., Ageshina A.A., Chesnokov G.A., Gribanov P.S., Topchiy M.A., Nechaev M.S., Asachenko A.F.
RSC Advances,
2019
35.

Ageshina A.A., Sterligov G.K., Rzhevskiy S.A., Topchiy M.A., Chesnokov G.A., Gribanov P.S., Melnikova E.K., Nechaev M.S., Asachenko A.F., Bermeshev M.V.
Dalton Transactions,
2019
36.

Topchiy M.A., Ageshina A.A., Chesnokov G.A., Sterligov G.K., Rzhevskiy S.A., Gribanov P.S., Osipov S.N., Nechaev M.S., Asachenko A.F.
ChemistrySelect,
2019
37.

Gribanov P.S., Atoian E.M., Philippova A.N., Topchiy M.A., Asachenko A.F., Osipov S.N.
European Journal of Organic Chemistry,
2021
38.
Rzhevskiy S.A., Topchiy M.A., Bogachev V.N., Ageshina A.A., Minaeva L.I., Sterligov G.K., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2021
39.
Rzhevskiy S.A., Topchiy M.A., Bogachev V.N., Minaeva L.I., Cherkashchenko I.R., Lavrov K.V., Sterligov G.K., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2021
40.

Chesnokov G.A., Ageshina A.A., Maryanova A.V., Rzhevskiy S.A., Gribanov P.S., Topchiy M.A., Nechaev M.S., Asachenko A.F.
Russian Chemical Bulletin,
2020
41.

Salvi L., Davis N.R., Ali S.Z., Buchwald S.L.
Organic Letters,
2011