Keywords
azomethine ylide
chiral substituent
diastereoselectivity
isatin
spiro compound
spirooxindole
thiohydantoin
Abstract
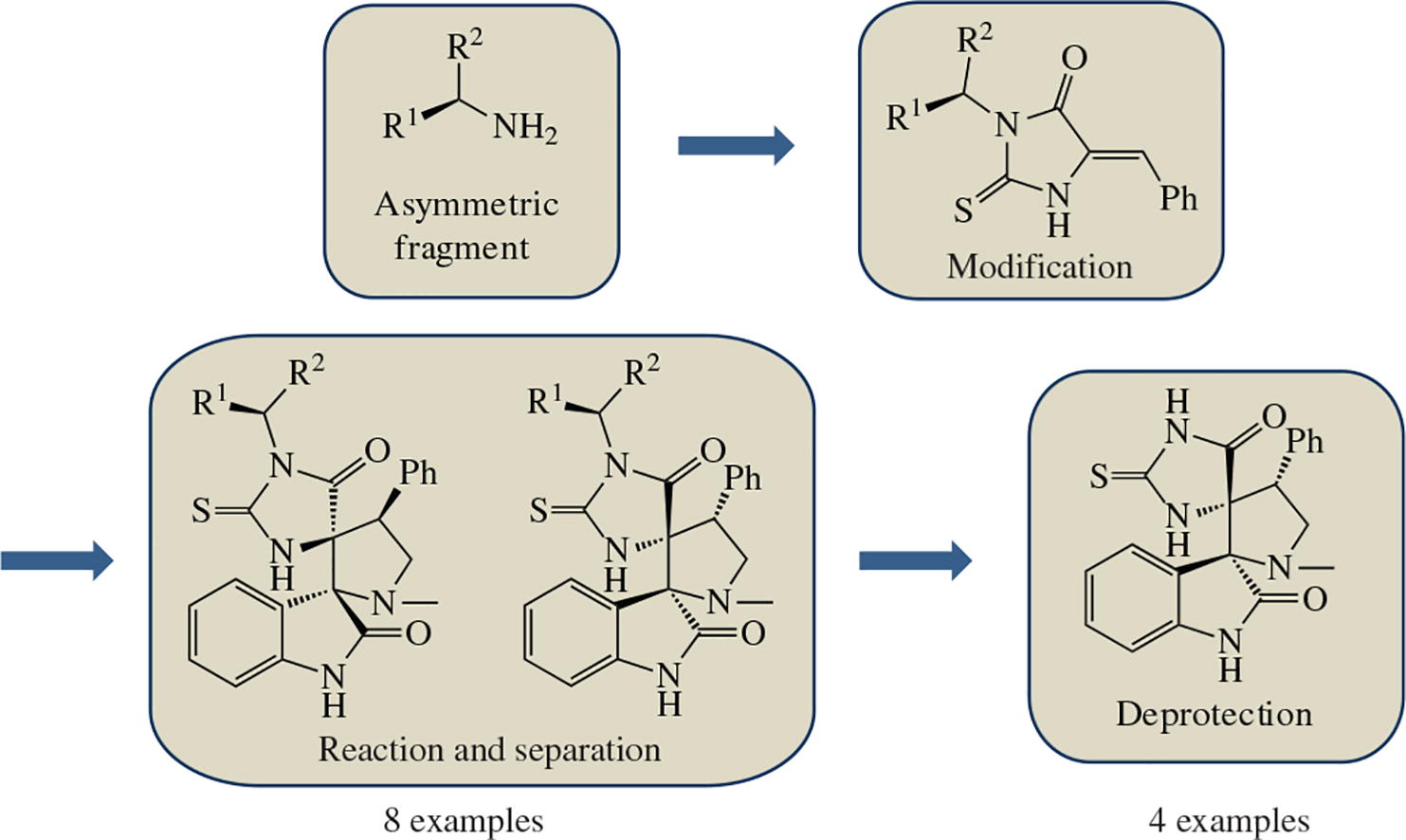
1,3-Dipolar cycloaddition of azomethine ylide, generated from isatin and sarcosine, with 5-arylidene-2-thiohydantoins equipped with various chiral substituents at the N3 atom occurs diastereoselectively. The highest selectivity with dr = 5 : 1 was observed for the 2-thiohydantoin bearing 1,2-diphenylethyl substituent.
References
1.

Wang S., Sun W., Zhao Y., McEachern D., Meaux I., Barrière C., Stuckey J.A., Meagher J.L., Bai L., Liu L., Hoffman-Luca C.G., Lu J., Shangary S., Yu S., Bernard D., et. al.
Cancer Research,
2014
2.

Shirai F., Mizutani A., Yashiroda Y., Tsumura T., Kano Y., Muramatsu Y., Chikada T., Yuki H., Niwa H., Sato S., Washizuka K., Koda Y., Mazaki Y., Jang M., Yoshida H., et. al.
Journal of Medicinal Chemistry,
2020
3.

Teja C., Babu S.N., Noor A., Daniel J.A., Devi S.A., Nawaz Khan F.R.
RSC Advances,
2020
4.

Zak K., Pecak A., Rys B., Wladyka B., Dömling A., Weber L., Holak T.A., Dubin G.
Expert Opinion on Therapeutic Patents,
2013
5.
![3-Nitro-2-phenyl-2-trifluoromethyl-2H-chromenes in reactions with azomethine ylides from isatins and (thia)proline: synthesis of spiro[chromeno(thia)pyrrolizidine-11,3'-oxindoles]](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kutyashev I.B., Kochnev I.А., Cherepkova A.А., Zimnitskiy N.S., Barkov A.Y., Korotaev V.Y., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2020
6.

Ivanenkov Y.A., Vasilevski S.V., Beloglazkina E.K., Kukushkin M.E., Machulkin A.E., Veselov M.S., Chufarova N.V., Chernyaginab E.S., Vanzcool A.S., Zyk N.V., Skvortsov D.A., Khutornenko A.A., Rusanov A.L., Tonevitsky A.G., Dontsova O.A., et. al.
Bioorganic and Medicinal Chemistry Letters,
2015
7.
![2-Azabicyclo[2.2.1]hept-5-en-3-one: chemical profile of a versatile synthetic building block and its impact on the development of therapeutics.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Singh R., Vince R.
Chemical Reviews,
2012
8.
Novotortsev V.K., Kukushkin M.E., Tafeenko V.A., Zyk N.V., Beloglazkina E.K.
Mendeleev Communications,
2020
9.

Kukushkin M.E., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Burmistrov V.V., Butov G.M., Zyk N.V., Majouga A.G., Beloglazkina E.K.
Phosphorus, Sulfur and Silicon and the Related Elements,
2020
10.

Beloglazkina A., Barashkin A., Polyakov V., Kotovsky G., Karpov N., Mefedova S., Zagribelny B., Ivanenkov Y., Kalinina M., Skvortsov D., Tafeenko V., Zyk N., Majouga A., Beloglazkina E.
Chemistry of Heterocyclic Compounds,
2020
11.

Krasnovskaya O.O., Malinnikov V.M., Dashkova N.S., Gerasimov V.M., Grishina I.V., Kireev I.I., Lavrushkina S.V., Panchenko P.A., Zakharko M.A., Ignatov P.A., Fedorova O.A., Jonusauskas G., Skvortsov D.A., Kovalev S.S., Beloglazkina E.K., et. al.
Bioconjugate Chemistry,
2019
12.

Ryczek J.
Journal of Heterocyclic Chemistry,
2003
13.

Davies S.G., Huckvale R., Lee J.A., Lorkin T.J., Roberts P.M., Thomson J.E.
Tetrahedron,
2012