Keywords
1,2,3-triazolium salts
1,2,4-triazolium salts
alkyltriazoles
dealkylation
nitrotriazoles
regioselectivity
SNipso-substitution
Abstract
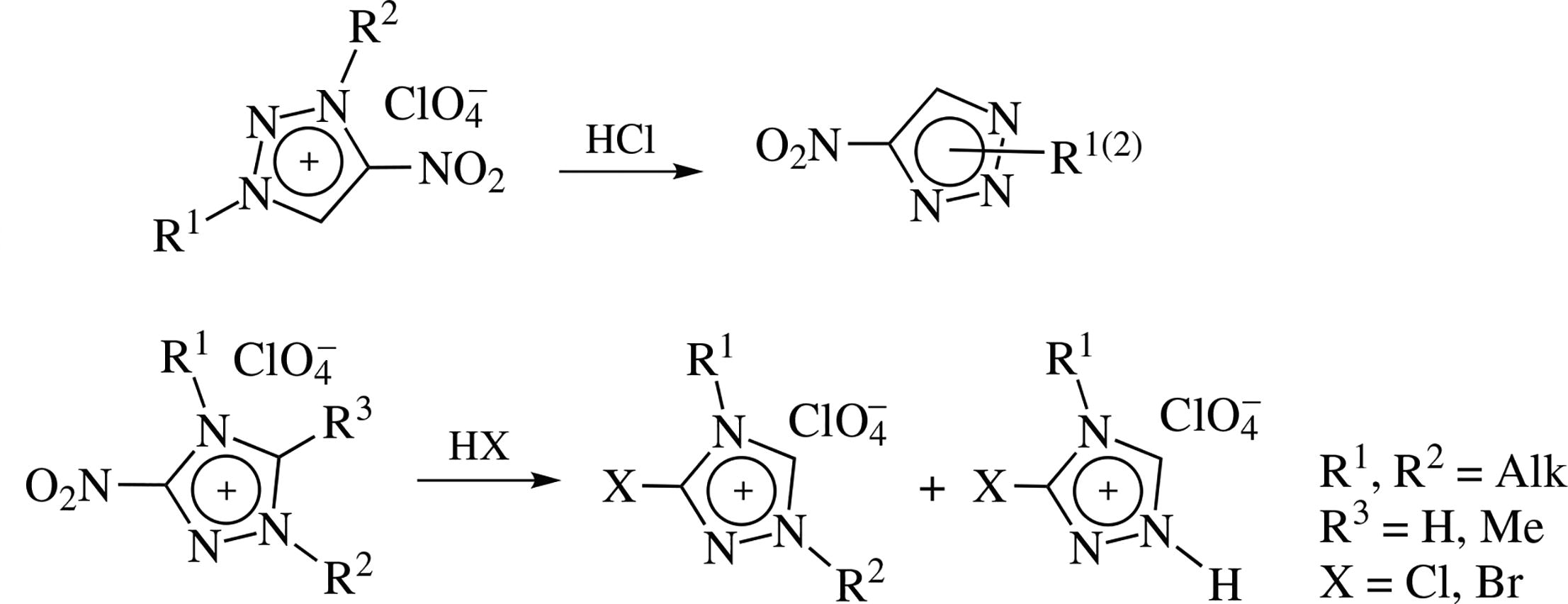
New selective synthesis of 1-alkyl-5-nitro-1,2,3-triazoles and 1-alkyl-4-nitro-1,2,3-triazoles has been developed, involving acid N-dealkylation of the relative 4-nitro-1,2,3- and 3-nitro- 5-R-1,2,4-triazolium salts. The assortment of novel 1-alkyl- 4(5)-nitro-1,2,3-triazoles has been thus essentially expanded. Treatment of relative 3-nitro-1,2,4-triazolium salts with HCl or HBr proceeds mostly as SNipso-substitution of the nitro group.
References
1.

Aggarwal R., Sumran G.
European Journal of Medicinal Chemistry,
2020
2.
(b) C. B. Vagish, P. Sudeep, H. P. Jayadevappaand and K. Ajay Kumar, Int. J. Curr. Res., 2020, 12, 12950
3.
(c) V. P. Krivopalov and O. P. Shkurko, Russ. Chem. Rev., 2005, 73, 339 (Usp. Khim., 2005, 73, 369)
4.

5.

Degtyarik M.M., Lyakhov A.S., Ivashkevich L.S., Voitekhovich S.V., Sukhanov G.T., Grigoriev Y.V.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2012
6.
Pasyukov D.V., Chernenko A.Y., Shepelenko K.E., Kutyrev V.V., Khrustalev V.N., Chernyshev V.M.
Mendeleev Communications,
2021
7.

Cai R., Yan W., Bologna M.G., de Silva K., Ma Z., Finklea H.O., Petersen J.L., Li M., Shi X.
Organic Chemistry Frontiers,
2015
8.
Topchiy M.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Khrustalev V.N., Paraschuk D.Y., Bermeshev M.V., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2019
9.
Topchiy M.A., Rzhevskiy S.A., Ageshina A.A., Kirilenko N.Y., Sterligov G.K., Mladentsev D.Y., Paraschuk D.Y., Osipov S.N., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2020
10.

Larina L., Lopyrev V.
2009
11.
(b) S. Gundala, M. R. Guda, A. F. Khasanov, D. S. Kopchuk, A. P. Krinochkin, S. Santra, G. V. Zyryanov, P. Venkatapuram, J. R. Garcia and V. N. Charushin, Mendeleev Commun., 2019, 29, 369
12.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019
13.
(d) E. G. Paronikyan, Sh. Sh. Dashyan and S. S. Mamyan, Mendeleev Commun., 2020, 30, 183.
14.
(a) R. W. Middleton, H. Monney and J. Parrick, Synthesis, 1984, 740; (b) W. Holzer, Tetrahedron, 1991, 47, 5471; (c) G. T. Sukhanov, Yu. V. Filippova and A. G. Sukhanova, Chem. Heterocycl. Compd., 2012, 48, 1340 (Khim. Geterotsikl. Soedin., 2012, 48, 1438); (d) Yu. V. Grigoriev, S. V. Voitekhovich and O. A. Ivashkevich, Russ. J. Org. Chem., 2012, 48, 610 (Zh. Org. Khim., 2012, 48, 611); (e) O. A. Ivashkevich, V. E. Matulis, P. N. Gaponik, G. T. Sukhanov, J. V. Filippova and A. G. Sukhanova, Chem. Heterocycl. Compd., 2008, 44, 1472 (Khim. Geterotsikl. Soedin., 2008, 44, 1816); (f) G. V. Sakovich, G. T. Sukhanov, Yu. V. Filippova, A. G. Sukhanova and K. K. Bosov, Russ. Chem. Bull., 2013, 62, 111.
15.
Sukhanov G.T., Sakovich G.V., Filippova Y.V., Bagryanskaya I.Y., Sukhanova A.G.
Mendeleev Communications,
2014
16.

Ivashkevich O.A., Matulis V.E., Lyakhov A.S., Grigorieva I.N., Gaponik P.N., Sukhanov G.T., Filippova Y.V., Sukhanova A.G.
Chemistry of Heterocyclic Compounds,
2009
17.

Filippova Y.V., Sukhanova A.G., Voitekhovich S.V., Matulis V.E., Sukhanov G.T., Grigoriev Y.V., Ivashkevich O.A.
Journal of Heterocyclic Chemistry,
2012
18.

Sukhanov G.T., Sukhanova A.G., Sheikov Y.V.
Chemistry of Heterocyclic Compounds,
2007
19.
10.1016/j.mencom.2022.03.020_b0045
Krupnova
Chem. Sustainable Dev.,
2017
20.

Bagal L.I., Pevzner M.S., Samarenko V.Y., Egorov A.P.
Chemistry of Heterocyclic Compounds,
1970
21.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
22.
10.1016/j.mencom.2022.03.020_b0060
SADABS
version 2008-1,
2008