Keywords
arylation
carboxylate assistance
CH-activation
N-heterocyclic carbene ligands
ruthenium complexes
Abstract
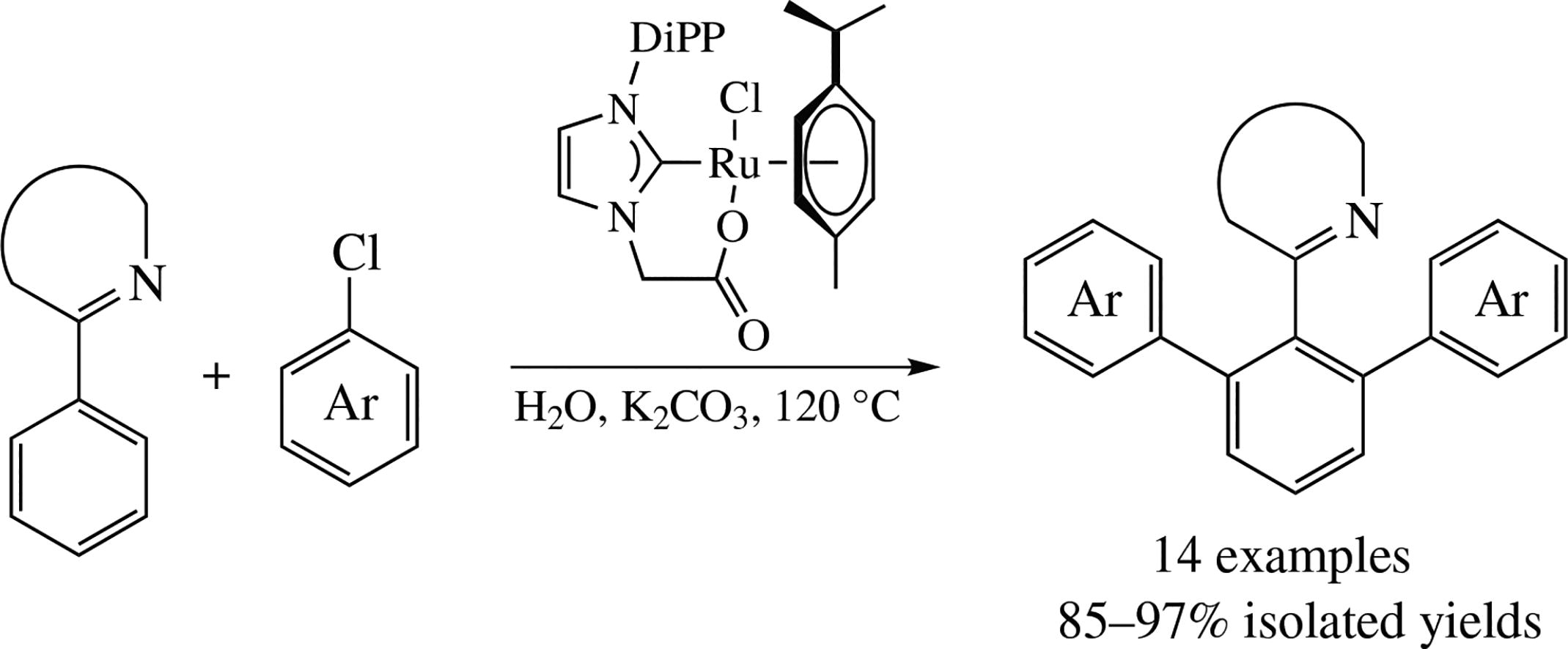
Ruthenium(ii) complexes with chelating N-heterocyclic carbene (NHC) ligands were studied in the arylation of phenyl group in 2-phenylpyridine and 1-phenylpyrazole with aryl chlorides in water. Complexes with NHC-ligands containing a hemilabile coordinating N-carboxylatomethyl group enable fast and selective ortho-CH-diarylation in the absence of carboxylate-assisting additives.
References
1.

Chu J.C., Rovis T.
Angewandte Chemie - International Edition,
2017
2.

Gandeepan P., Ackermann L.
Chem,
2018
3.

Gandeepan P., Müller T., Zell D., Cera G., Warratz S., Ackermann L.
Chemical Reviews,
2018
4.

Seki M.
Organic Process Research and Development,
2016
5.

Cernak T., Dykstra K.D., Tyagarajan S., Vachal P., Krska S.W.
Chemical Society Reviews,
2016
6.
10.1016/j.mencom.2022.03.017_b0030
Shome
Chem. Rec.,
1935
7.

Zha G., Qin H., Kantchev E.A.
RSC Advances,
2016
8.

Nareddy P., Jordan F., Szostak M.
ACS Catalysis,
2017
9.
10.1016/j.mencom.2022.03.017_b0045
Zhao
Chem. Rev.,
1981
10.

Rufino-Felipe E., Nayely Osorio-Yáñez R., Vera M., Valdés H., González-Sebastián L., Reyes-Sanchez A., Morales-Morales D.
Polyhedron,
2021
11.

Shepelenko K.E., Soliev S.B., Galushko A.S., Chernyshev V.M., Ananikov V.P.
Inorganic Chemistry Frontiers,
2021
12.

Khazipov O.V., Shepelenko K.E., Pasyukov D.V., Chesnokov V.V., Soliev S.B., Chernyshev V.M., Ananikov V.P.
Organic Chemistry Frontiers,
2021
13.
Cabanes J., Odnoroh M., Duhayon C., Bijani C., Sournia-Saquet A., Polia R., Labande A.
Mendeleev Communications,
2021
14.

Özdemir I., Demir S., Çetinkaya B., Gourlaouen C., Maseras F., Bruneau C., Dixneuf P.H.
Journal of the American Chemical Society,
2008
15.

Yaşar S., Doǧan Ö., Özdemir I., Çetinkaya B.
Applied Organometallic Chemistry,
2008
16.

Demir S., Özdemir I., Çetinkaya B.
Journal of Organometallic Chemistry,
2009
17.
10.1016/j.mencom.2022.03.017_b0085
Özdemir
Eur. J. Inorg. Chem.,
2009
18.
10.1016/j.mencom.2022.03.017_b0090
Demir
Synlett,
2010
19.

Prades A., Poyatos M., Peris E.
Advanced Synthesis and Catalysis,
2010
20.

Sabater S., Mata J.A., Peris E.
Organometallics,
2012
21.

Gonell S., Peris E.
ACS Catalysis,
2014
22.

Kaloğlu N., Özdemir İ., Gürbüz N., Arslan H., Dixneuf P.
Molecules,
2018
23.

Ackermann L., Vicente R., Potukuchi H.K., Pirovano V.
Organic Letters,
2010
24.

Diers E., Phani Kumar N.Y., Mejuch T., Marek I., Ackermann L.
Tetrahedron,
2013
25.

Hubrich J., Himmler T., Rodefeld L., Ackermann L.
ACS Catalysis,
2015
26.

Zhang J., Yang Q., Zhu Z., Yuan M.L., Fu H.Y., Zheng X.L., Chen H., Li R.X.
European Journal of Organic Chemistry,
2012
27.

Ackermann L.
Chemical Reviews,
2011
28.

Fabre I., von Wolff N., Le Duc G., Ferrer Flegeau E., Bruneau C., Dixneuf P.H., Jutand A.
Chemistry - A European Journal,
2013
29.

Shan C., Zhu L., Qu L., Bai R., Lan Y.
Chemical Society Reviews,
2018
30.

Chernyshev V.M., Denisova E.A., Eremin D.B., Ananikov V.P.
Chemical Science,
2020
31.

Chernyshev V.M., Khazipov O.V., Eremin D.B., Denisova E.A., Ananikov V.P.
Coordination Chemistry Reviews,
2021
32.

Gandolfi C., Heckenroth M., Neels A., Laurenczy G., Albrecht M.
Organometallics,
2009
33.

Ledoux N., Allaert B., Verpoort F.
European Journal of Inorganic Chemistry,
2007
34.

DePasquale J., White N.J., Ennis E.J., Zeller M., Foley J.P., Papish E.T.
Polyhedron,
2013
35.

Mangalum A., McMillen C.D., Tennyson A.G.
Inorganica Chimica Acta,
2015
36.

Arockiam P.B., Fischmeister C., Bruneau C., Dixneuf P.H.
Green Chemistry,
2013
37.

Binnani C., Rai R.K., Tyagi D., Mobin S.M., Singh S.K.
European Journal of Inorganic Chemistry,
2018
38.
Bruker APEX-III, Bruker AXS Inc., Madison, WI, 2019.
39.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
40.
CrysAlisPro, Version 1.171.41.106a, Rigaku Oxford Diffraction, 2021.
41.
10.1016/j.mencom.2022.03.017_b0205
Sheldrick
Acta Crystallogr.,
2015
42.
10.1016/j.mencom.2022.03.017_b0210
Sheldrick
Acta Crystallogr.,
2015
43.

Spek A.L.
Acta crystallographica. Section C, Structural chemistry,
2015