Keywords
Acetate
Amantadine
Bismuth
Iodide
perovskite photovoltaics
Abstract
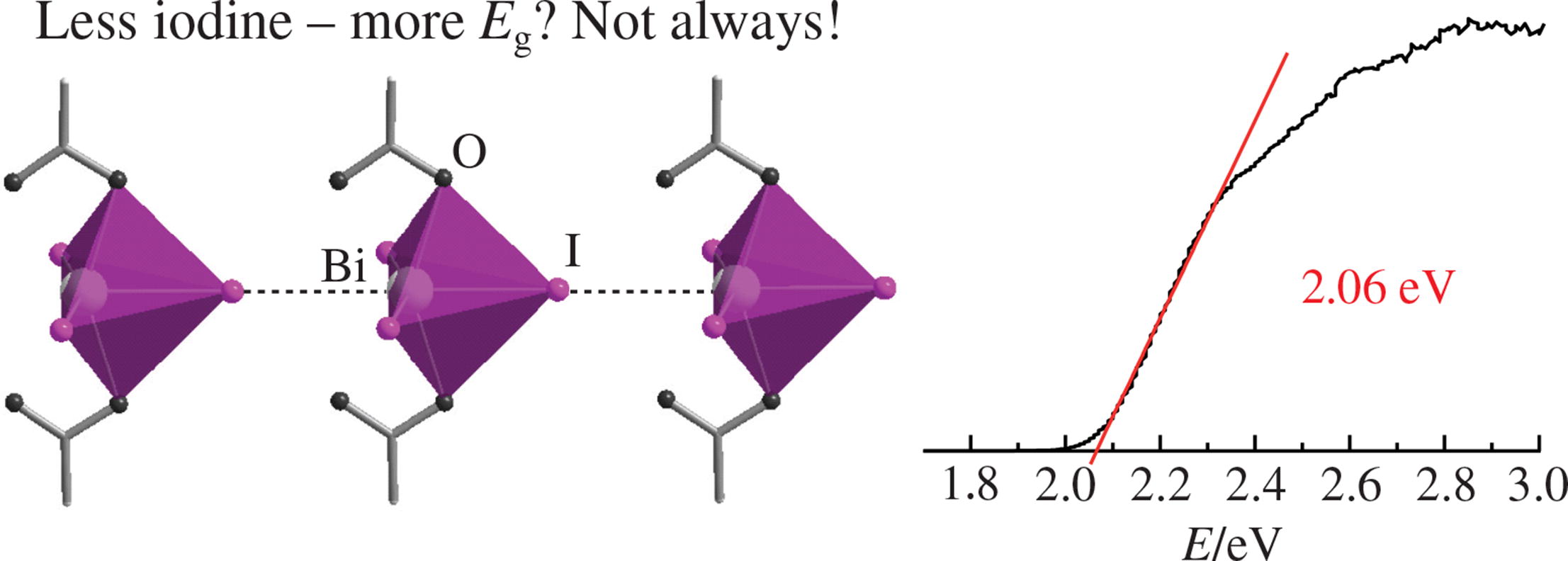
The amantadinium iodoacetatobismuthate(III) [C10H15NH3·(CH3)2CO]2[BiI3.67(CH3COO)1.33] is a new hybrid halometallate with iodide ions partially replaced by oxygen-containing acetates to form stronger interaction between the anionic and cationic substructures. The title compound as well-shaped orange-red crystals was synthesized by a facile reaction in acetone solution in the presence of glacial acetic acid. The crystal structure of the compound consists of the infinite anionic chains [BiI3.67(CH3COO)1.33]2– and the countercations [C10H15NH3·(CH3)2CO]+; according to the optical absorption data, the test compound is a semiconductor with a band gap of 2.06 eV.
References
1.

Lin K., Xing J., Quan L.N., de Arquer F.P., Gong X., Lu J., Xie L., Zhao W., Zhang D., Yan C., Li W., Liu X., Lu Y., Kirman J., Sargent E.H., et. al.
Nature,
2018
2.

Shen Y., Cheng L., Li Y., Li W., Chen J., Lee S., Tang J.
Advanced Materials,
2019
3.

Cao Y., Wang N., Tian H., Guo J., Wei Y., Chen H., Miao Y., Zou W., Pan K., He Y., Cao H., Ke Y., Xu M., Wang Y., Yang M., et. al.
Nature,
2018
4.

Sahli F., Werner J., Kamino B.A., Bräuninger M., Monnard R., Paviet-Salomon B., Barraud L., Ding L., Diaz Leon J.J., Sacchetto D., Cattaneo G., Despeisse M., Boccard M., Nicolay S., Jeangros Q., et. al.
Nature Materials,
2018
5.

27.9% Efficient Monolithic Perovskite/Silicon Tandem Solar Cells on Industry Compatible Bottom Cells
Köhnen E., Wagner P., Lang F., Cruz A., Li B., Roß M., Jošt M., Morales-Vilches A.B., Topič M., Stolterfoht M., Neher D., Korte L., Rech B., Schlatmann R., Stannowski B., et. al.
Solar RRL,
2021
6.

Cao F., Li L.
Advanced Functional Materials,
2020
7.

Li T., Hu Y., Morrison C.A., Wu W., Han H., Robertson N.
Sustainable Energy and Fuels,
2017
8.
J. Zhang, S. Han, C. Ji, W. Zhang, Y. Wang, K. Tao, Z. Sun, J. Luo, Chem. – Eur. J. 23 (2017) 17304. Z. Zhang, X. Li, X. Xia, Z. Wang, Z. Huang, B. Lei, Y. Gao, J. Phys. Chem. Lett. 8 (2017) 4300.
9.
![Polymeric iodobismuthates {[Bi3I10]} and {[BiI4]} with N-heterocyclic cations: promising perovskite-like photoactive materials for electronic devices](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Usoltsev A.N., Elshobaki M., Adonin S.A., Frolova L.A., Derzhavskaya T., Abramov P.A., Anokhin D.V., Korolkov I.V., Luchkin S.Y., Dremova N.N., Stevenson K.J., Sokolov M.N., Fedin V.P., Troshin P.A.
Journal of Materials Chemistry A,
2019
10.

Adonin S.A., Sokolov M.N., Fedin V.P.
Coordination Chemistry Reviews,
2016
11.

Adonin S.A., Sokolov M.N., Fedin V.P.
Coordination Chemistry Reviews,
2018
12.

Shestimerova T.A., Mironov A.V., Bykov M.A., Starichenkova E.D., Kuznetsov A.N., Grigorieva A.V., Shevelkov A.V.
Crystal Growth and Design,
2019
13.

Shestimerova T.A., Golubev N.A., Yelavik N.A., Bykov M.A., Grigorieva A.V., Wei Z., Dikarev E.V., Shevelkov A.V.
Crystal Growth and Design,
2018
14.
Kotov V.Y., Ilyukhin A.B., Sadovnikov A.A., Birin K.P., Simonenko N.P., Nguyen H.T., Baranchikov A.E., Kozyukhin S.A.
Mendeleev Communications,
2017
15.

Liebing P., Stein F., Hilfert L., Lorenz V., Oliynyk K., Edelmann F.T.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2018
16.

Dennington A.J., Weller M.T.
Dalton Transactions,
2016
17.

Adonin S.A., Gorokh I.D., Novikov A.S., Samsonenko D.G., Yushina I.V., Sokolov M.N., Fedin V.P.
CrystEngComm,
2018
18.

Li T., Wang Q., Nichol G.S., Morrison C.A., Han H., Hu Y., Robertson N.
Dalton Transactions,
2018
19.

Shestimerova T.A., Mironov A.V., Bykov M.A., Grigorieva A.V., Wei Z., Dikarev E.V., Shevelkov A.V.
Molecules,
2020
20.
![Supramolecular organization of the organic-inorganic hybrid [{p-(CH3)2NH—C6H4—NH3}2Cl][BiI6]: assembly of a three-dimensional structure via covalent and non-covalent interactions](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shestimerova T.A., Golubev N.A., Grigorieva A.V., Bykov M.A., Wei Z., Dikarev E.V., Shevelkov A.V.
Russian Chemical Bulletin,
2021
21.

Shui Q., Tang H., Fu R., Fang Y., Ma Z., Wu X.
Angewandte Chemie - International Edition,
2020
22.

Daub M., Hillebrecht H.
Angewandte Chemie - International Edition,
2015
23.
10.1016/j.mencom.2022.03.014_b0115
SAINT
Data Reduction and Correction Program, version 8.38A,
2017
24.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
25.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
26.
![The motley family of polar compounds (MV)[M(X5−xX′x)] based on anionic chains of trans-connected M(III)(X,X′)6 octahedra (M=Bi, Sb; X, X′=Cl, Br, I) and methylviologen (MV) dications](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Leblanc N., Mercier N., Allain M., Toma O., Auban-Senzier P., Pasquier C.
Journal of Solid State Chemistry,
2012
27.
![Large Spontaneous Polarization and Clear Hysteresis Loop of a Room-Temperature Hybrid Ferroelectric Based on Mixed-Halide [BiI3Cl2] Polar Chains and Methylviologen Dication](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Leblanc N., Mercier N., Zorina L., Simonov S., Auban-Senzier P., Pasquier C.
Journal of the American Chemical Society,
2011
28.
![[NH2(C2H4)2O]MX5: a new family of morpholinium nonlinear optical materials among halogenoantimonate(iii) and halogenobismuthate(iii) compounds. Structural characterization, dielectric and piezoelectric properties](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Owczarek M., Szklarz P., Jakubas R., Miniewicz A.
Dalton Transactions,
2012
29.

Bi W., Leblanc N., Mercier N., Auban-Senzier P., Pasquier C.
Chemistry of Materials,
2009
30.

Adonin S.A., Gorokh I.D., Novikov A.S., Abramov P.A., Sokolov M.N., Fedin V.P.
Chemistry - A European Journal,
2017
31.
Kotov V.Y., Ilyukhin A.B., Buikin P.A., Yorov K.E.
Mendeleev Communications,
2019
32.

Chen Y., Yang Z., Guo C., Ni C., Ren Z., Li H., Lang J.
European Journal of Inorganic Chemistry,
2010
33.

Kotov V.Y., Ilyukhin A.B., Korlyukov A.A., Smol’yakov A.F., Kozyukhin S.A.
New Journal of Chemistry,
2018
34.
Stålhandske C., Kjekshus A., Svensson S., Holme T., Lindberg A.A., Jansen G., Lamm B., Samuelsson B.
Acta Chemica Scandinavica,
1969
35.

Kelly A.W., Wheaton A.M., Nicholas A.D., Patterson H.H., Pike R.D.
Journal of Inorganic and Organometallic Polymers and Materials,
2018
36.

Baranov A.I., Kloo L., Olenev A.V., Popovkin B.A., Romanenko A.I., Shevelkov A.V.
Journal of the American Chemical Society,
2001
37.
10.1016/j.mencom.2022.03.014_b0185
Yu
Eur. J. Inorg. Chem.,
1989
38.
![Dynamic Directing Effect and Symmetric Correlation in Three pH‐Modulated 1,4‐Diazabicyclo[2.2.2]octane/Iodoargentate Hybrids](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Yu T., Li H., Hao P., Shen J., Fu Y.
European Journal of Inorganic Chemistry,
2016
39.
![Trinuclear iodobismuthate complex [Na3(Me2CO)12][Bi3I12]: Synthesis and crystal structure](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Adonin S.A., Peresypkina E.V., Sokolov M.N., Fedin V.P.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2014
40.

Yelovik N.A., Mironov A.V., Bykov M.A., Kuznetsov A.N., Grigorieva A.V., Wei Z., Dikarev E.V., Shevelkov A.V.
Inorganic Chemistry,
2016
41.
![Crystal structure, vibrational and optical properties of a new self-organized material containing iodide anions of bismuth(III), [C6H4(NH3)2]2Bi2I10·4H2O](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Hrizi C., Samet A., Abid Y., Chaabouni S., Fliyou M., Koumina A.
Journal of Molecular Structure,
2011
42.

Laane J., Jagodzinski P.W.
Inorganic Chemistry,
1980
43.
10.1016/j.mencom.2022.03.014_b0215
Mousdis
B: J. Chem. Sci.,
1998