Keywords
3-diones
aldehydes
cross-conjugated cyclopentene-1
cyclopentenones
cytotoxicity
Friedel–Crafts reaction
ketals
Knoevenagel condensation
organochlorine compounds
Suzuki–Miyaura reaction
Abstract
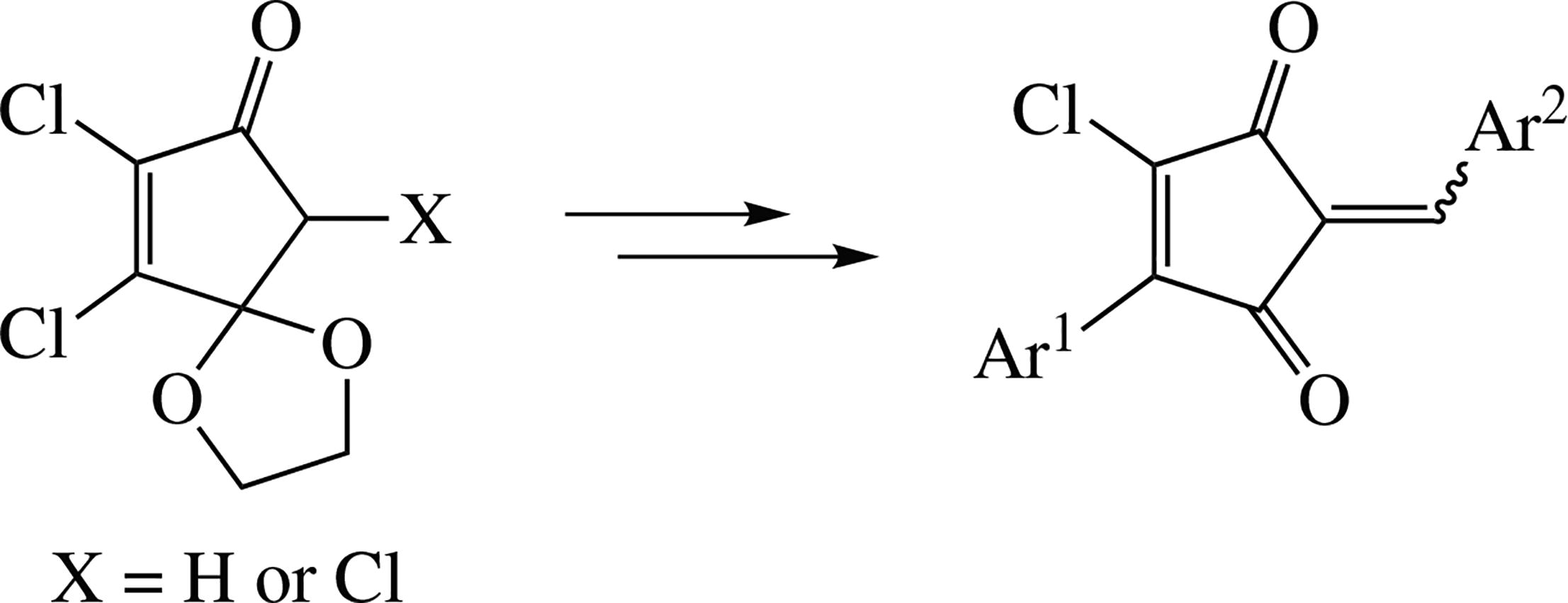
Novel 2,4-dichloro-5-phenylcyclopent-4-ene-1,3-dione and 5-aryl-4-chlorocyclopent-4-ene-1,3-dione were synthesized by the Suzuki–Miyaura or Friedel–Crafts reactions of the di- and trichlorocyclopentenone monoketals and subsequent hydrolysis of the ketal function. Condensation of these diones with (hetero)aromatic aldehydes afforded multifunctional 1,3-cyclopentenediones that showed anticancer activity.Keywords: organochlorine compounds, cyclopentenones, ketals, Suzuki–Miyaura reaction, Friedel–Crafts reaction, Knoevenagel condensation, aldehydes, cross-conjugated cyclopentene-1,3-diones, cytotoxicity.
References
1.

Li X., Ferreira D., Jacob M.R., Zhang Q., Khan S.I., ElSohly H.N., Nagle D.G., Smillie T.J., Khan I.A., Walker L.A., Clark A.M.
Journal of the American Chemical Society,
2004
2.

Hori H.
Bioorganic and Medicinal Chemistry,
2002
3.

Babu K.S., Li X., Jacob M.R., Zhang Q., Khan S.I., Ferreira D., Clark A.M.
Journal of Medicinal Chemistry,
2006
4.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
5.

Loza V.V., Gimazetdinov A.M., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2018
6.
10.1016/j.mencom.2022.03.010_b0030
Takahashi
J. Chem. Soc., Chem. Commun.,
1982
7.

Akhmetvaleev R.R., Akbutina F.A., Ivanova N.A., Miftakhov M.S.
Russian Chemical Bulletin,
2001
8.

Kuznetsov O.M., Akhmetvateev R.R., Vostrikov N.S., Miftakhov M.S.
Russian Chemical Bulletin,
1996
9.

Miyaura N., Suzuki A.
Chemical Reviews,
1995
10.
(b) A. Suzuki, Angew. Chem., Int. Ed., 2011, 50, 6723
11.

Suzuki A.
Journal of Organometallic Chemistry,
1999
12.

Rossi R., Bellina F., Carpita A.
Synthesis,
2004
13.
Vasil’ev A.A., Burukin A.S., Zhdankina G.M., Zlotin S.G.
Mendeleev Communications,
2021
14.
Matyugina E.S., Khandazhinskaya A.L., Kochetkov S.N., Seley-Radtke K.L.
Mendeleev Communications,
2020
15.
Nosova E.V., Moshkina T.N., Lipunova G.N., Baklanova I.V., Kopchuk D.S., Slepukhin P.A., Charushin V.N.
Mendeleev Communications,
2018
16.

Khera R.A., Hussain M., Hung N.T., Eleya N., Feist H., Villinger A., Langer P.
Helvetica Chimica Acta,
2012
17.

Hussain M., Hung N.T., Khera R.A., Villinger A., Langer P.
Tetrahedron Letters,
2011
18.

Zinad D.S., Hussain M., Villinger A., Langer P.
European Journal of Organic Chemistry,
2011
19.
Egorov V.A., Gimalova F.A., Zileeva Z.R., Zainullina L.F., Vakhitova Y.V., Miftakhov M.S.
Mendeleev Communications,
2019
20.

Egorov V.A., Khalilov L.M., Mescheryakova E.S., Gimalova F.A., Miftakhov M.S.
Russian Chemical Bulletin,
2021
21.

Williamson R.T., Buevich A.V., Martin G.E., Parella T.
Journal of Organic Chemistry,
2014
22.

Bigler P., Furrer J.
Magnetic Resonance in Chemistry,
2018
23.

Bigler P., Furrer J.
Magnetic Resonance in Chemistry,
2019