Keywords
1-Dioxide
1,4,2,6-Dithiadiazinane 1
catalysis
Cyclothiomethylation
cytotoxic activity
Hydrogen sulfide
Samarium compounds
Sodium sulfide
Sulfamide
Ytterbium compounds
Abstract
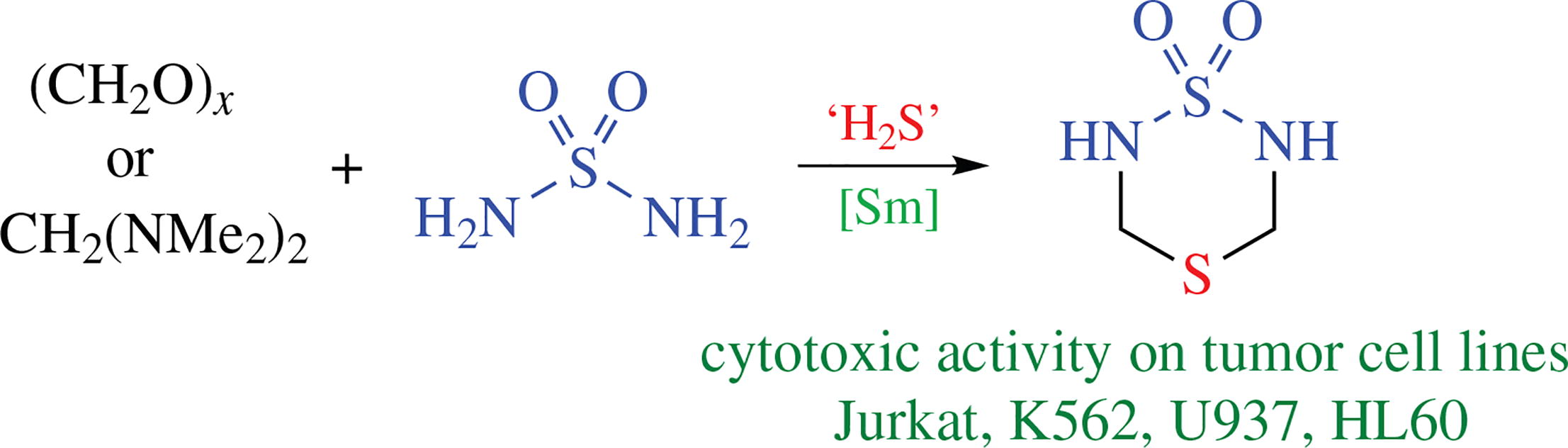
Cyclothiomethylation of sulfamide using paraformaldehyde or bis(dimethylamino)methane, hydrogen sulfide or its sodium salts catalyzed by rare-earth salts affords 1,4,2,6-dithiadi azinane 1,1-dioxide. The heterocycle was found to exhibit a pronounced cytotoxic effect against suspension tumor cell lines (Jurkat, HL60, K562, and U937).
References
1.

Rakhimova E.B., Kirsanov V.Y., Mescheryakova E.S., Khalilov L.M., Ibragimov A.G., Dzhemileva L.U., D’yakonov V.A., Dzhemilev U.M.
ACS Medicinal Chemistry Letters,
2019
2.
10.1016/j.mencom.2022.03.008_b0010
Makhmudiyarova
Molecules,
1874
3.
Evdokimova A.V., Alexeev A.A., Nurieva E.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2021
4.

Khairullina R.R., Tyumkina T.V., Akhmetshina G.A., Abdullin M.F., Ibragimov A.G.
Russian Chemical Bulletin,
2021
5.

Khairullina R.R., Geniyatova A.R., Tyumkina T.V., Karamzina D.S., Ibragimov A.G., Dzhemilev U.M.
Tetrahedron,
2017
6.

R. Akhmetova V., R. Nadyrgulova G., T. Niatshina Z., R. Khairullina R., A. Starikova Z., O. Borisova A., Yu. Antipin M., V. Kunakova R., M. Dzhemilev U.
Heterocycles,
2009
7.

Gazieva G.A., Kravchenko A.N., Trunova N.S., Makhova N.N.
Russian Chemical Bulletin,
2007
8.

Badgujar J.R., More D.H., Meshram J.S.
Indian Journal of Microbiology,
2017
9.

Vullo D., Innocenti A., Nishimori I., Pastorek J., Scozzafava A., Pastoreková S., Supuran C.T.
Bioorganic and Medicinal Chemistry Letters,
2005
10.

Vullo D., Voipio J., Innocenti A., Rivera C., Ranki H., Scozzafava A., Kaila K., Supuran C.T.
Bioorganic and Medicinal Chemistry Letters,
2005
11.

Berrino E., Bua S., Mori M., Botta M., Murthy V.S., Vijayakumar V., Tamboli Y., Bartolucci G., Mugelli A., Cerbai E., Supuran C.T., Carta F.
Molecules,
2017
12.
10.1016/j.mencom.2022.03.008_b0060
Ross
Patent EP 0104611,
1985
13.

Tait A., Luppi A., Franchini S., Preziosi E., Parenti C., Buccioni M., Marucci G., Leonardi A., Poggesi E., Brasili L.
Bioorganic and Medicinal Chemistry Letters,
2005
14.

Tait A., Luppi A., Hatzelmann A., Fossa P., Mosti L.
Bioorganic and Medicinal Chemistry,
2005
15.

Pearson R.G.
Journal of Chemical Education,
1968