Keywords
Anti-proliferative agents
Cell viability
Diazo coupling
Enzyme inhibitors
Michael acceptors
MTT assay
Rhodium catalysis
sulfonamides
Abstract
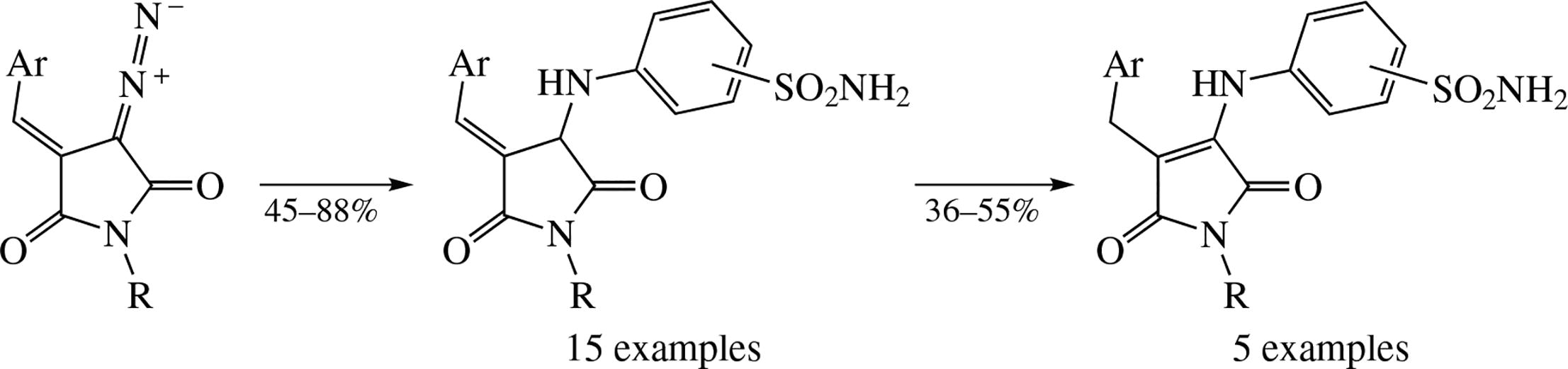
The earlier described (E)-3-arylidene-4-diazopyrrolidine-2,5-diones have been elaborated into two distinct series of compounds both bearing a primary sulfonamide moiety and an electrophilic ‘Michael acceptor’ motif. These compounds demonstrated cytotoxicity against colorectal cancer cell line HCT 116.
References
1.

Sharonova T., Paramonova P., Kalinin S., Bunev A., Gasanov R.Е., Nocentini A., Sharoyko V., Tennikova T.B., Dar’in D., Supuran C.T., Krasavin M.
European Journal of Medicinal Chemistry,
2021
2.
Chupakhin E.G., Kantin G.P., Dar’in D.V., Krasavin M.
Mendeleev Communications,
2021
3.

Yuan H., Hong K., Liu X., Qian Y., Xu X., Hu W.
Chemical Communications,
2021
4.

Nakano Y., Hamaguchi M., Nagai T.
Journal of Organic Chemistry,
1989
5.

Hamaguchi M., Misumi T., Oshima T.
Tetrahedron Letters,
1998
6.

Mboge M., Mahon B., McKenna R., Frost S.
Metabolites,
2018
7.

Gan F., Kaminska K.K., Yang H., Liew C., Leow P., So C., Tu L.N., Roy A., Yap C., Kang T., Chui W., Chew E.
Antioxidants and Redox Signaling,
2013
8.

Kaminska K.K., Bertrand H.C., Tajima H., Stafford W.C., Cheng Q., Chen W., Wells G., Arner E.S., Chew E.
Oncotarget,
2016
9.

Krasavin M., Sharonova T., Sharoyko V., Zhukovsky D., Kalinin S., Žalubovskis R., Tennikova T., Supuran C.T.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2020
10.

Rajput A., Dominguez San Martin I., Rose R., Beko A., LeVea C., Sharratt E., Mazurchuk R., Hoffman R.M., Brattain M.G., Wang J.
Journal of Surgical Research,
2008