Keywords
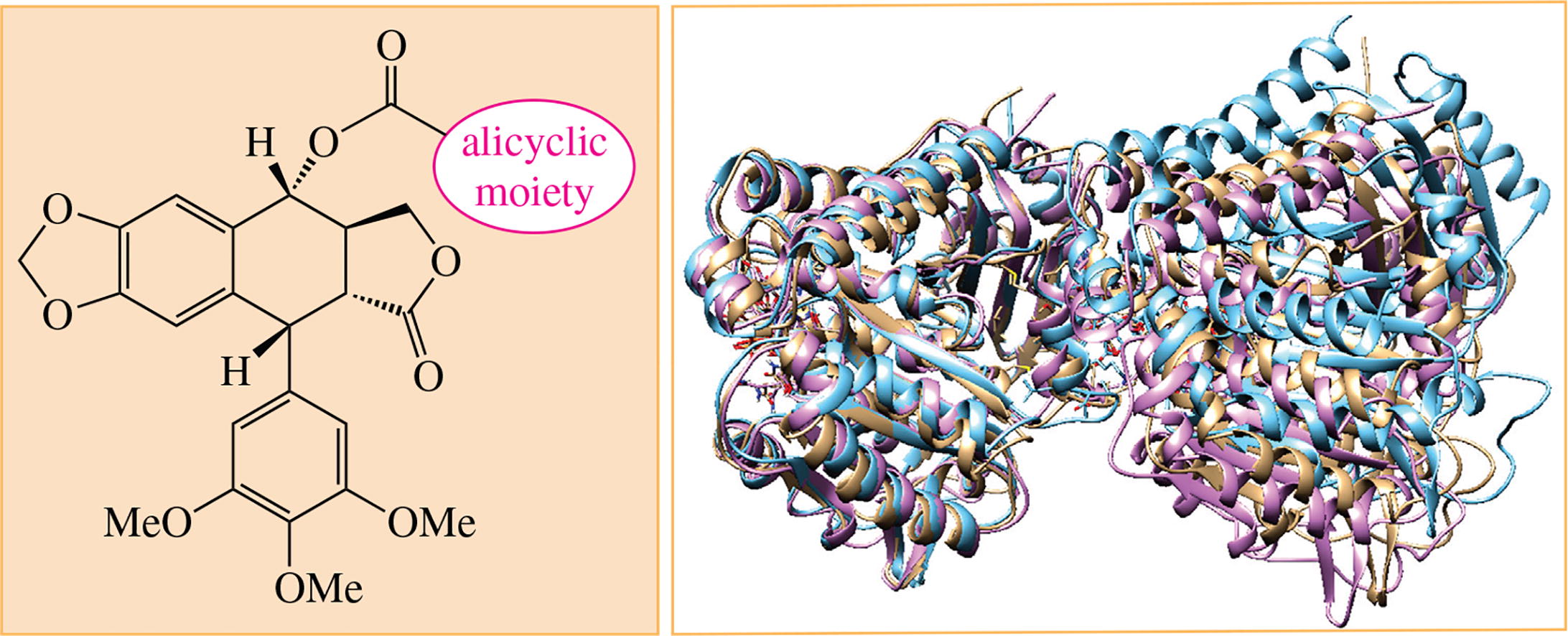
alicyclic compounds
Bridged moieties
esters
Human lung carcinoma A549
Immunofluorescence microscopy
Microtubules
molecular dynamics
Podophyllotoxin
tubulin
Abstract
Immunofluorescent microscopy of cancer cells A549 treated with novel alicyclic (mostly bridged) podophyllotoxin C4-esters at different concentrations gave evidence that the ‘curling’ of microtubules occurred at one of the first steps of their depolymerisation. Molecular dynamics study revealed the differences in curved conformations of tubulin dimer in a complex with adamantane-comprising ester and in a complex with podophyllotoxin.
References
1.

Cheng Z., Lu X., Feng B.
Translational Cancer Research,
2020
2.

Naaz F., Haider M.R., Shafi S., Yar M.S.
European Journal of Medicinal Chemistry,
2019
3.

La Regina G., Coluccia A., Naccarato V., Silvestri R.
European Journal of Pharmaceutical Sciences,
2019
4.

Yu X., Che Z., Xu H.
Chemistry - A European Journal,
2017
5.

Zhang X., Rakesh K.P., Shantharam C.S., Manukumar H.M., Asiri A.M., Marwani H.M., Qin H.
Bioorganic and Medicinal Chemistry,
2018
6.

Liu Y., Tian J., Qian K., Zhao X., Morris-Natschke S.L., Yang L., Nan X., Tian X., Lee K.
Medicinal Research Reviews,
2014
7.

Han H., Lin H., He D., Ren Y., Sun W., Liang L., Du M., Li D., Chu Y., Yang M., Wang X., Yang Y.
Chemistry and Biodiversity,
2018
8.

Wu G., Xu B., Yang Y., Zhang X., Fang K., Ma T., Wang H., Xue N., Chen M., Guo W., Jia X., Wang P., Lei H.
European Journal of Medicinal Chemistry,
2018
9.

Zhao W., He L., Xiang T., Tang Y.
European Journal of Medicinal Chemistry,
2019
10.
10.1016/j.mencom.2022.03.006_b0050
Wei
Front. Chem.,
2019
11.

Sun W., Ji Y., Wan Y., Han H., Lin H., Lu G., Qi J., Wang X., Yang Y.
Bioorganic and Medicinal Chemistry Letters,
2017
12.
Zefirov N.A., Kruth A., Wobith B., Nurieva E.V., Riyaz S., Reddy C.V., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2018
13.

Zefirov N.A., Evteeva Y.A., Wobith B., Kuznetsov S.A., Zefirova O.N.
Structural Chemistry,
2018
14.

López-Pérez J.L., del Olmo E., de Pascual-Teresa B., Abad A., San Feliciano A.
Bioorganic and Medicinal Chemistry Letters,
2004
15.
![Podophyllotoxin analogue with bicyclo[3.2.1]octane moiety annelated with indole: synthesis, molecular modeling, and biological testing](/storage/images/resized/ORapGEvQagyY4uwGVkDHuIo7YUCJvWzb7YLElWuZ_small_thumb.webp)
Zefirov N.A., Lavrushkina E.A., Kuznetsov S.A., Zefirova O.N.
Biomeditsinskaya Khimiya,
2019
16.

Gerlier D., Thomasset N.
Journal of Immunological Methods,
1986
17.

Mosmann T.
Journal of Immunological Methods,
1983
18.
Zefirov N.A., Gadert L., Fatkulin A.R., Shibilev V.M., Butov G.M., Mokhov V.M., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
19.

Nurieva E.V., Zefirov N.A., Temnyakova N.S., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2020
20.

Lavrushkina E.A., Shibilev V.M., Zefirov N.A., Shevtsova E.F., Shevtsov P.N., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2020
21.

Al-Haddad A., Shonn M.A., Redlich B., Blocker A., Burkhardt J.K., Yu H., Hammer J.A., Weiss D.G., Steffen W., Griffiths G., Kuznetsov S.A.
Molecular Biology of the Cell,
2001
22.

23.
Nurieva E.V., Zefirov N.A., Fritsch N., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
24.

Zefirov N.A., Mamaeva A.V., Krasnoperova A.I., Evteeva Y.A., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2021
25.
Zefirov N.A., Evteeva Y.A., Krasnoperova A.I., Mamaeva A.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
26.

Schilstra M.J., Martin S.R., Bayley P.M.
Journal of Biological Chemistry,
1989
27.
10.1016/j.mencom.2022.03.006_b0135
Prota
J. Mol. Biol.,
1848
28.

Ravelli R.B., Gigant B., Curmi P.A., Jourdain I., Lachkar S., Sobel A., Knossow M.
Nature,
2004
29.

Dorléans A., Gigant B., Ravelli R.B., Mailliet P., Mikol V., Knossow M.
Proceedings of the National Academy of Sciences of the United States of America,
2009
30.

Barbier P., Dorléans A., Devred F., Sanz L., Allegro D., Alfonso C., Knossow M., Peyrot V., Andreu J.M.
Journal of Biological Chemistry,
2010
31.

Pettersen E.F., Goddard T.D., Huang C.C., Couch G.S., Greenblatt D.M., Meng E.C., Ferrin T.E.
Journal of Computational Chemistry,
2004
32.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
33.

Huang J., MacKerell A.D.
Journal of Computational Chemistry,
2013
34.

Lee J., Cheng X., Swails J.M., Yeom M.S., Eastman P.K., Lemkul J.A., Wei S., Buckner J., Jeong J.C., Qi Y., Jo S., Pande V.S., Case D.A., Brooks C.L., MacKerell A.D., et. al.
Journal of Chemical Theory and Computation,
2015