Keywords
(Thiophen-2-yl)cyclopropane dicarboxylate
donor–acceptor cyclopropanes
Gallium salts
Ipso-dimerization
Pentaleno[6a,1-b]-thiophenes
Abstract
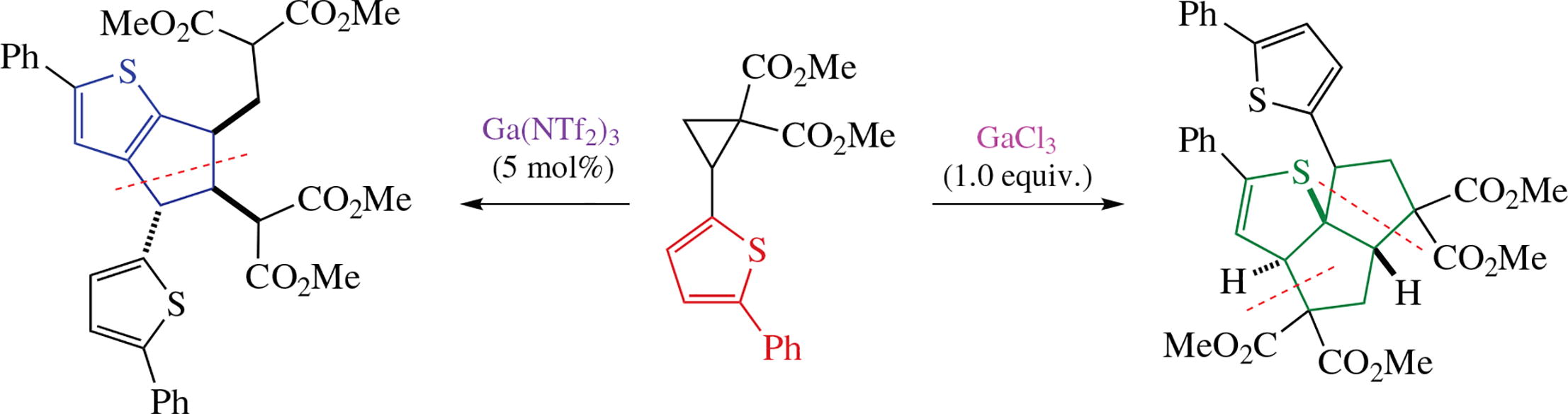
A selectivity-switched dimerization process for (5-phenyl-2-thienyl)cyclopropane-1,1-dicarboxylate under activation conditions with anhydrous Gaiii salts was developed. Using GaCl3 or Ga(NTf2)3 as catalysts, 3a,4,5a,6,7,8-hexahydro-5H-pentaleno[6a,1-b]thiophene and 5,6-dihydro-4H-cyclo-penta[b]thiophene derivatives can be selectively obtained as a result of ipso-type and [3+2]-annulation type dimerization. The crucial role of a phenyl substituent at position 5 of the thiophene ring and some regularities are discussed.
References
1.

Reissig H., Zimmer R.
Chemical Reviews,
2003
2.

Schneider T.F., Kaschel J., Werz D.B.
Angewandte Chemie - International Edition,
2014
3.

Tomilov Y.V., Menchikov L.G., Novikov R.A., Ivanova O.A., Trushkov I.V.
Russian Chemical Reviews,
2018
4.

Pirenne V., Muriel B., Waser J.
Chemical Reviews,
2020
5.

Augustin A.U., Werz D.B.
Accounts of Chemical Research,
2021
6.

Kolb S., Petzold M., Brandt F., Jones P.G., Jacob C.R., Werz D.B.
Angewandte Chemie - International Edition,
2021
7.

Andreev I.A., Ratmanova N.K., Augustin A.U., Ivanova O.A., Levina I.I., Khrustalev V.N., Werz D.B., Trushkov I.V.
Angewandte Chemie - International Edition,
2021
8.

Ortega A., Uria U., Tejero T., Prieto L., Reyes E., Merino P., Vicario J.L.
Organic Letters,
2021
9.
Novikov R.A., Tomilov Y.V.
Mendeleev Communications,
2015
10.
![Cascade Cleavage of Three-Membered Rings in the Reaction of D–A Cyclopropanes with 4,5-Diazaspiro[2.4]hept-4-enes: A Route to Highly Functionalized Pyrazolines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Novikov R.A., Borisov D.D., Zotova M.A., Denisov D.A., Tkachev Y.V., Korolev V.A., Shulishov E.V., Tomilov Y.V.
Journal of Organic Chemistry,
2018
11.
![Highly diastereoselective formation of 3,7-dioxabicyclo[3.3.0]octan-2-ones in reaction of 2-arylcyclopropanedicarboxylates with aromatic aldehydes using 1,2-zwitterionic reactivity type](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Borisov D.D., Novikov R.A., Tomilov Y.V.
Tetrahedron Letters,
2017
12.
![Domino Cyclodimerization of Indole-Derived Donor-Acceptor Cyclopropanes: One-Step Construction of the Pentaleno[1,6-a,b]indole Skeleton](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ivanova O.A., Budynina E.M., Chagarovskiy A.O., Rakhmankulov E.R., Trushkov I.V., Semeykin A.V., Shimanovskii N.L., Melnikov M.Y.
Chemistry - A European Journal,
2011
13.

Ivanova O.A., Budynina E.M., Chagarovskiy A.O., Trushkov I.V., Melnikov M.Y.
Journal of Organic Chemistry,
2011
14.
![Dimerization of Dimethyl 2-(Naphthalen-1-yl)cyclopropane-1,1-dicarboxylate in the Presence of GaCl3to [3+2], [3+3], [3+4], and Spiroannulation Products](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Novikov R.A., Tomilov Y.V.
Helvetica Chimica Acta,
2013
15.

Mondal B., Das D., Saha J.
Organic Letters,
2020
16.

Jacob A., Jones P.G., Werz D.B.
Organic Letters,
2020
17.

Chagarovskiy A.O., Budynina E.M., Ivanova O.A., Grishin Y.K., Trushkov I.V., Verteletskii P.V.
Tetrahedron,
2009
18.
![[3+2] Cyclodimerization of 2-arylcyclopropane-1,1-diesters. Lewis acid induced reversion of cyclopropane umpolung](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chagarovskiy A.O., Ivanova O.A., Budynina E.M., Trushkov I.V., Melnikov M.Y.
Tetrahedron Letters,
2011
19.

Ivanova O.A., Budynina E.M., Skvortsov D.A., Limoge M., Bakin A.V., Chagarovskiy A.O., Trushkov I.V., Melnikov M.Y.
Chemical Communications,
2013
20.
![A straightforward approach to tetrahydroindolo[3,2-b]carbazoles and 1-indolyltetrahydrocarbazoles through [3+3] cyclodimerization of indole-derived cyclopropanes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ivanova O.A., Budynina E.M., Khrustalev V.N., Skvortsov D.A., Trushkov I.V., Melnikov M.Y.
Chemistry - A European Journal,
2015
21.

Novikov R.A., Borisov D.D., Tarasova A.V., Tkachev Y.V., Tomilov Y.V.
Angewandte Chemie - International Edition,
2018
22.

Novikov R.A., Denisov D.A., Potapov K.V., Tkachev Y.V., Shulishov E.V., Tomilov Y.V.
Journal of the American Chemical Society,
2018
23.

Borisov D.D., Novikov R.A., Tomilov Y.V.
Angewandte Chemie - International Edition,
2016
24.
![GaCl3-Mediated Cascade [2 + 4]-Cycloaddition/[4 + 2]-Annulation of Donor–Acceptor Cyclopropanes with Conjugated Dienes: Strategy for the Construction of Benzobicyclo[3.3.1]nonane Skeleton](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Belaya M.A., Knyazev D.A., Borisov D.D., Novikov R.A., Tomilov Y.V.
Journal of Organic Chemistry,
2021
25.
![Shortcut Approach to Cyclopenta[b]indoles by [3+2] Cyclodimerization of Indole-Derived Cyclopropanes](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Shortcut Approach to Cyclopenta[b]indoles by [3+2] Cyclodimerization of Indole-Derived Cyclopropanes
Budynina E., Ivanova O., Skvortsov D., Trushkov I., Melnikov M.
Synlett,
2014
26.

Tomilov Y.V., Novikov R.A., Borisov D.D., Chermashentsev G.R.
Synthesis,
2021
27.
10.1016/j.mencom.2022.03.005_b0135
Zhu
Org. Lett.,
1856
28.
![Synthesis of Indenopyridine Derivatives via MgI2‐Promoted [2+4] Cycloaddition Reaction of In‐situ Generated 2‐Styrylmalonate from Donor‐Acceptor Cyclopropanes and Chalconimines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Verma K., Banerjee P.
Advanced Synthesis and Catalysis,
2018
29.

Borisov D.D., Chermashentsev G.R., Novikov R.A., Tomilov Y.V.
Journal of Organic Chemistry,
2021