Keywords
N-hydroxyphthalimide
N-oxyl radicals
free radicals
hypervalent iodine
imides
vinyl azides
Abstract
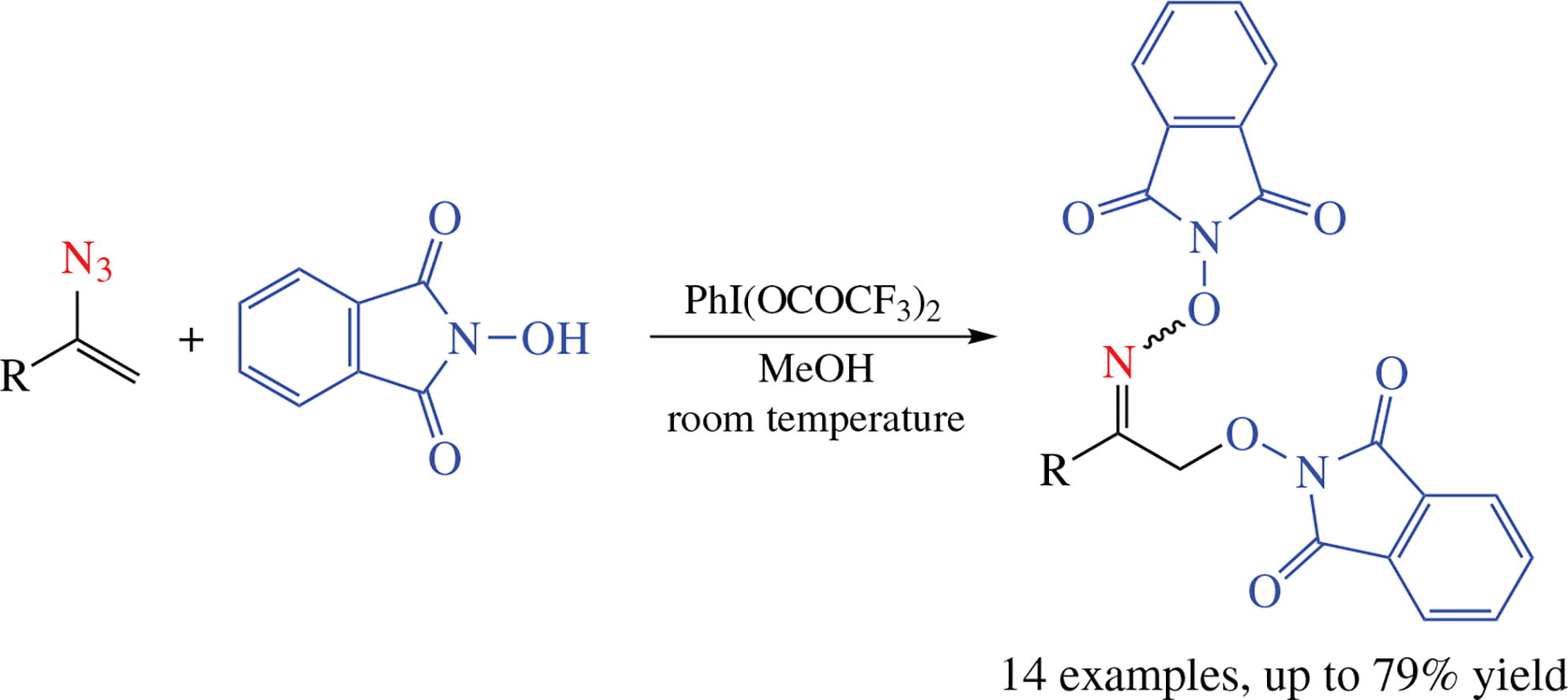
O,O′-Bis(phthalimido)-modified 2-(hydroxyimino)ethanols containing N–O–N fragment were synthesized in high yields via the reaction of vinyl azides with N-hydroxyphthalimide under the action of hypervalent iodine-based oxidant. The reaction proceeds under mild conditions and is compatible with a wide range of vinyl azides. Presumably, the process starts with the oxidative formation of phthalimide-N-oxyl radical, followed by its addition to vinyl azide with the subsequent trapping of the generated iminyl radical with the second phthalimide-N-oxyl radical.
References
1.

A versatile strategy for difunctionalization of carbon–carbon multiple bonds by photoredox catalysis
Koike T., Akita M.
Organic Chemistry Frontiers,
2016
2.

Yin G., Mu X., Liu G.
Accounts of Chemical Research,
2016
3.

Lin L., Liang Q., Kong X., Chen Q., Xu B.
Journal of Organic Chemistry,
2020
4.

Wu Z., Hu M., Li J., Wu W., Jiang H.
Organic and Biomolecular Chemistry,
2021
5.

Fu X., Zhao W.
Chinese Journal of Organic Chemistry,
2019
6.

Bag R., De P.B., Pradhan S., Punniyamurthy T.
European Journal of Organic Chemistry,
2017
7.

Tang S., Liu K., Liu C., Lei A.
Chemical Society Reviews,
2015
8.

Lan X., Wang N., Xing Y.
European Journal of Organic Chemistry,
2017
9.

Wu X., Wu S., Zhu C.
Tetrahedron Letters,
2018
10.

Yao H., Hu W., Zhang W.
Molecules,
2020
11.

Li Z., Fang G., Gu Q., Liu X.
Chemical Society Reviews,
2020
12.

Bao X., Li J., Jiang W., Huo C.
Synthesis,
2019
13.

Hu B., DiMagno S.G.
Organic and Biomolecular Chemistry,
2015
14.

Hayashi H., Kaga A., Chiba S.
Journal of Organic Chemistry,
2017
15.

Fu J., Zanoni G., Anderson E.A., Bi X.
Chemical Society Reviews,
2017
16.
(a) Q. Liang, L. Lin, G. Li, X. Kong and B. Xu, Chin. J. Chem., 2021, 39, 1948; (b) O. M. Mulina, N. V. Zhironkina, S. A. Paveliev, D. V. Demchuk and A. O. Terent’ev, Org. Lett., 2020, 22, 1818; (c) Y. Liao, Y. Ran, G. Liu, P. Liu and X. Liu, Org. Chem. Front., 2020, 7, 3638; (d) J. Jang and D. Y. Kim, Bull. Korean Chem. Soc., 2020, 41, 370; (e) H. I. Jung and D. Y. Kim, Synth. Commun., 2020, 50, 380; (f) Y. Yao, Z. Yin, W. Chen, W. Xie, F.-S. He and J. Wu, Adv. Synth. Catal., 2021, 363, 570; (g) Y.-Z. Pan, S.-Y. Cheng, Q.-Y. Li, H.-T. Tang, Y.-M. Pan, X.-J. Meng and Z.-Y. Mo, Synlett, 2021, 32, 593; (h) M. Luo, X. Ren, R. Shen, C. Qi, Y. Zhang and H. Wang, ChemistrySelect, 2021, 6, 2471; (i) P.-F. Zhong, H.-M. Lin, L.-W. Wang, Z.-Y. Mo, X.-J. Meng, H.-T. Tang and Y.-M. Pan, Green Chem., 2020, 22, 6334; (j) G. Li, X. Kong, Q. Liang, L. Lin, K. Yu, B. Xu and Q. Chen, Eur. J. Org. Chem., 2020, 6135; (k) O. M. Mulina, A. I. Ilovaisky, T. Opatz and A. O. Terent’ev, Tetrahedron Lett., 2021, 64, 152737.
17.

Paveliev S.A., Alimkhanova L.S., Sergeeva A.V., Terent'ev A.O.
Tetrahedron Letters,
2020
18.

Paveliev S.A., Churakov A.I., Alimkhanova L.S., Segida O.O., Nikishin G.I., Terent'ev A.O.
Advanced Synthesis and Catalysis,
2020
19.

Krylov I.B., Paveliev S.A., Syroeshkin M.A., Korlyukov A.A., Dorovatovskii P.V., Zubavichus Y.V., Nikishin G.I., Terent’ev A.O.
Beilstein Journal of Organic Chemistry,
2018
20.

Krylov I.B., Paveliev S.A., Matveeva O.K., Terent'ev A.O.
Tetrahedron,
2019