Keywords
2-thiohydantoins
amino acids
exocyclic C=C bond
heterocyclization
isothiocyanates
Abstract
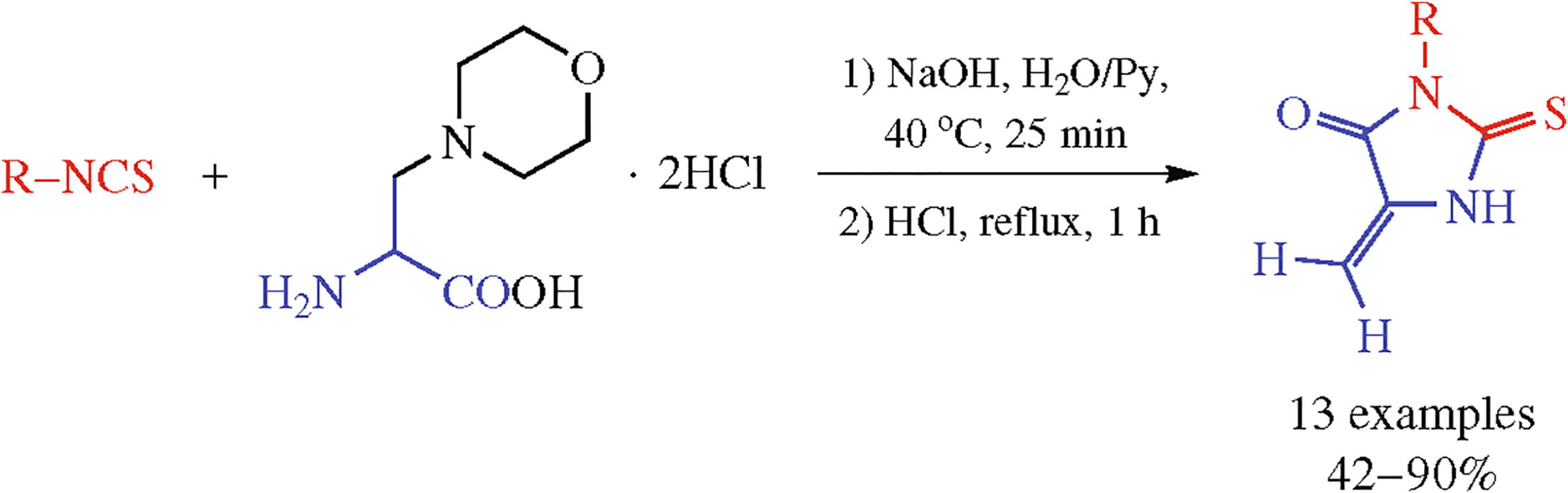
3-Aryl-5-methylidene-2-thiohydantoins were constructed in one-pot reaction of aryl isothiocyanates and 3-morpholino- alanine in alkaline medium with the subsequent treatment with boiling hydrochloric acid.
References
1.

Cho S., Kim S., Shin D.
European Journal of Medicinal Chemistry,
2019
2.

Konnert L., Lamaty F., Martinez J., Colacino E.
Chemical Reviews,
2017
3.
10.1016/j.mencom.2022.01.041_b0015
Hahn
J. Am. Chem. Soc.,
1927
4.
10.1016/j.mencom.2022.01.041_b0020
McKee
J. Am. Chem. Soc.,
2020
5.

Litzinger A.
Journal of the American Chemical Society,
1934
6.

Antipin R.L., Beloglazkina E.K., Magouga A.G., Chernysheva A.N., Zyk N.V.
Chemistry of Heterocyclic Compounds,
2008
7.

Hassaneen H.M., Daboun H.A., Abdelhadi H.A., Abdel-reheim N.A.
Phosphorus, Sulfur and Silicon and the Related Elements,
1995
8.
![First example of [3+2] cycloaddition of azomethine ylides to 5-methylidene-3-phenylhydantoin](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kukushkin M.E., Kondratyeva A.A., Zyk N.V., Majouga A.G., Beloglazkina E.K.
Russian Chemical Bulletin,
2019
9.
Shybanov D.E., Kukushkin M.E., Tafeenko V.A., Zyk N.V., Grishin Y.K., Roznyatovsky V.A., Beloglazkina E.K.
Mendeleev Communications,
2021
10.

Kuznetsova O.Y., Antipin R.L., Udina A.V., Krasnovskaya O.O., Beloglazkina E.K., Terenin V.I., Koteliansky V.E., Zyk N.V., Majouga A.G.
Journal of Heterocyclic Chemistry,
2015
11.

Ding M., Sun Y., Liu X., Liu Z.
Organic Preparations and Procedures International,
2003
12.

Khodair A.I.
Carbohydrate Research,
2001
13.

Kukushkin M.E., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Burmistrov V.V., Butov G.M., Zyk N.V., Majouga A.G., Beloglazkina E.K.
Phosphorus, Sulfur and Silicon and the Related Elements,
2020
14.

Evdokimov N.M., Magedov I.V., McBrayer D., Kornienko A.
Bioorganic and Medicinal Chemistry Letters,
2016
15.

Sumoto K., Fujisaki F., Shoji K.
Heterocycles,
2009
16.

Atropisomerism-Induced Facial Selectivity in Nitrile Oxide Cycloadditions with 5-Methylenehydantoins
Said A.M., Savage G.P.
Journal of Organic Chemistry,
2011
17.

Shih H., Cheng W.
Tetrahedron Letters,
2008
18.

Fujisaki F., Aki H., Naito A., Fukami E., Kashige N., Miake F., Sumoto K.
Chemical and Pharmaceutical Bulletin,
2014
19.

L. Gleason J., A. Cernak T.
Heterocycles,
2007
20.

Fujisaki F., Shoji K., Sumoto K.
Chemical and Pharmaceutical Bulletin,
2009
21.

Galun A.B.
Journal of Chemical & Engineering Data,
1976
22.

ABE N., FUJISAKI F., SUMOTO K.
Chemical and Pharmaceutical Bulletin,
2011
23.
Edman P., Dynesen E., Webb M., Rottenberg M.
Acta Chemica Scandinavica,
1950
24.

Yakusheva A.V., Galibeev S.S., Spiridonova R.R., Samuilov Y.D., Kochnev A.M.
Polymer Science - Series B,
2008