Keywords
o-diarylazoles
antimitotic activity
isoxazoles
microtubule destabilization
nitrostilbenes
pyrazoles
pyrroles
sea urchin embryo
triazoles
Abstract
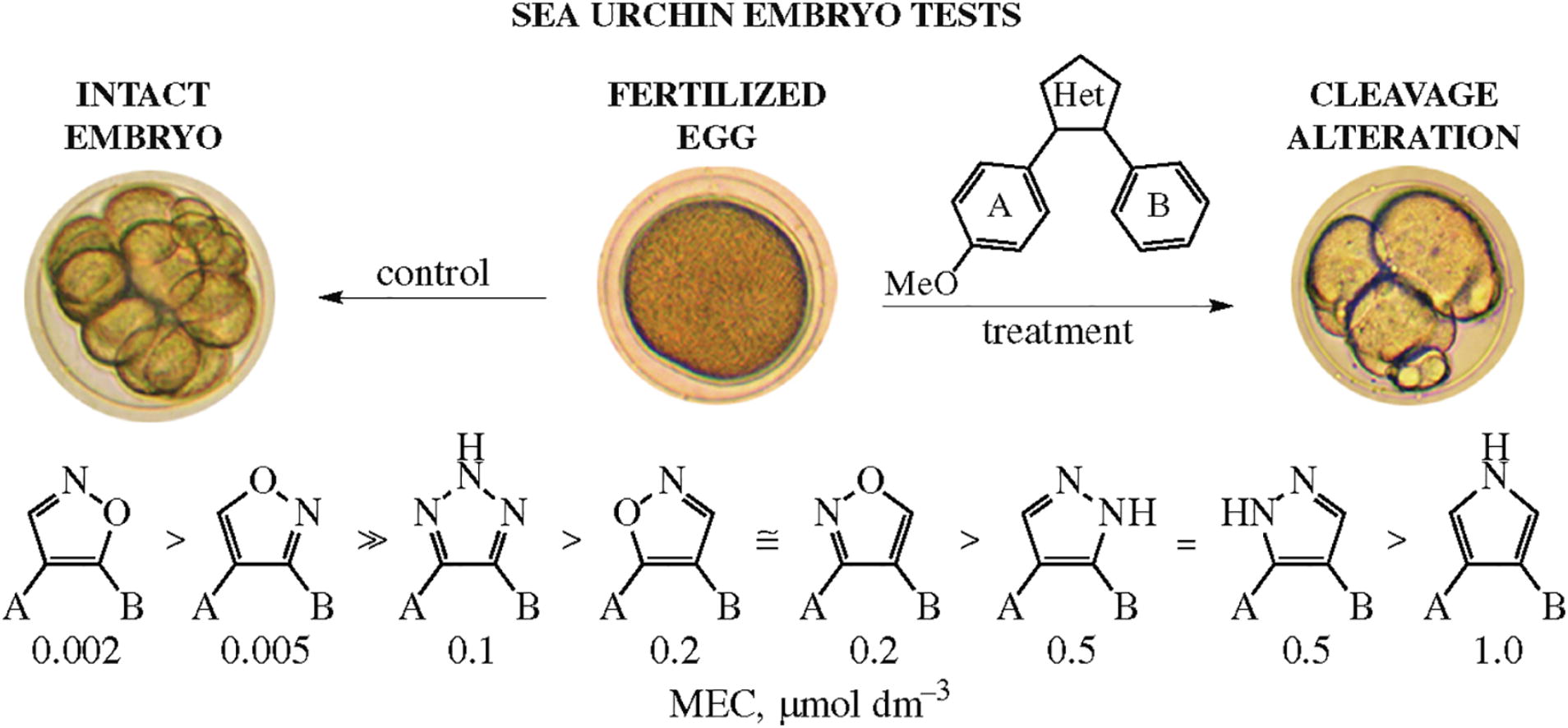
Monomethoxy substituted o-diarylazoles with isoxazole, triazole, pyrazole, or pyrrole linkers were synthesized and evaluated for antimitotic antitubulin activity in a sea urchin embryo model. Structure–activity relationship study revealed that only isoxazole heterocycle together with the unsubstituted phenyl ring next to the heteroatom endowed the molecule with appropriate configuration to exhibit antiproliferative effect by microtubule destabilizing mode of action.
References
1.

Dumontet C., Jordan M.A.
Nature Reviews Drug Discovery,
2010
2.

Duan Y., Liu W., Tian L., Mao Y., Song C.
Current Topics in Medicinal Chemistry,
2019
3.

Naaz F., Haider M.R., Shafi S., Yar M.S.
European Journal of Medicinal Chemistry,
2019
4.

Jordan A., Hadfield J.A., Lawrence N.J., McGown A.T.
Medicinal Research Reviews,
1998
5.

Negi A.S., Gautam Y., Alam S., Chanda D., Luqman S., Sarkar J., Khan F., Konwar R.
Bioorganic and Medicinal Chemistry,
2015
6.

Dong M., Liu F., Zhou H., Zhai S., Yan B.
Molecules,
2016
7.

Li L., Jiang S., Li X., Liu Y., Su J., Chen J.
European Journal of Medicinal Chemistry,
2018
8.

Pati H.N., Wicks M., Holt Jr. H.L., LeBlanc R., Weisbruch P., Forrest L., Lee M.
Heterocyclic Communications,
2005
9.

Cafici L., Pirali T., Condorelli F., Del Grosso E., Massarotti A., Sorba G., Canonico P.L., Tron G.C., Genazzani A.A.
Journal of Combinatorial Chemistry,
2008
10.

Liu T., Dong X., Xue N., Wu R., He Q., Yang B., Hu Y.
Bioorganic and Medicinal Chemistry,
2009
11.

Burja B., Čimbora-Zovko T., Tomić S., Jelušić T., Kočevar M., Polanc S., Osmak M.
Bioorganic and Medicinal Chemistry,
2010
12.

Semenova M.N., Demchuk D.V., Tsyganov D.V., Chernysheva N.B., Samet A.V., Silyanova E.A., Kislyi V.P., Maksimenko A.S., Varakutin A.E., Konyushkin L.D., Raihstat M.M., Kiselyov A.S., Semenov V.V.
ACS Combinatorial Science,
2018
13.

Gaukroger K., Hadfield J.A., Lawrence N.J., Nolan S., McGown A.T.
Organic and Biomolecular Chemistry,
2003
14.

Semenova M.N., Kiselyov A.S., Titov I.Y., Raihstat M.M., Molodtsov M., Grishchuk E., Spiridonov I., Semenov V.V.
Chemical Biology and Drug Design,
2007
15.

Semenova M.N., Kiselyov A.S., Tsyganov D.V., Konyushkin L.D., Firgang S.I., Semenov R.V., Malyshev O.R., Raihstat M.M., Fuchs F., Stielow A., Lantow M., Philchenkov A.A., Zavelevich M.P., Zefirov N.S., Kuznetsov S.A., et. al.
Journal of Medicinal Chemistry,
2011
16.

Shestopalov A.M., Litvinov Y.M., Rodinovskaya L.A., Malyshev O.R., Semenova M.N., Semenov V.V.
ACS Combinatorial Science,
2012
17.

Semenova M.N., Tsyganov D.V., Malyshev O.R., Ershov O.V., Bardasov I.N., Semenov R.V., Kiselyov A.S., Semenov V.V.
Bioorganic and Medicinal Chemistry Letters,
2014
18.
![Synthesis and anti-mitotic activity of 6,7-dihydro-4H-isothiazolo[4,5-b]pyridin-5-ones: In vivo and cell-based studies](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Semenov V.V., Lichitsky B.V., Komogortsev A.N., Dudinov A.A., Krayushkin M.M., Konyushkin L.D., Atamanenko O.P., Karmanova I.B., Strelenko Y.A., Shor B., Semenova M.N., Kiselyov A.S.
European Journal of Medicinal Chemistry,
2017
19.

Stroylov V.S., Svitanko I.V., Maksimenko A.S., Kislyi V.P., Semenova M.N., Semenov V.V.
Bioorganic and Medicinal Chemistry Letters,
2020
20.

Chernysheva N.B., Maksimenko A.S., Andreyanov F.A., Kislyi V.P., Strelenko Y.A., Khrustalev V.N., Semenova M.N., Semenov V.V.
European Journal of Medicinal Chemistry,
2018
21.

Quan X., Ren Z., Wang Y., Guan Z.
Organic Letters,
2014
22.

Silyanova E.A., Samet A.V., Semenov V.V.
Russian Chemical Bulletin,
2018