Keywords
cascade reaction
energetic compound
furazan
furoxan
fused pyridine
Abstract
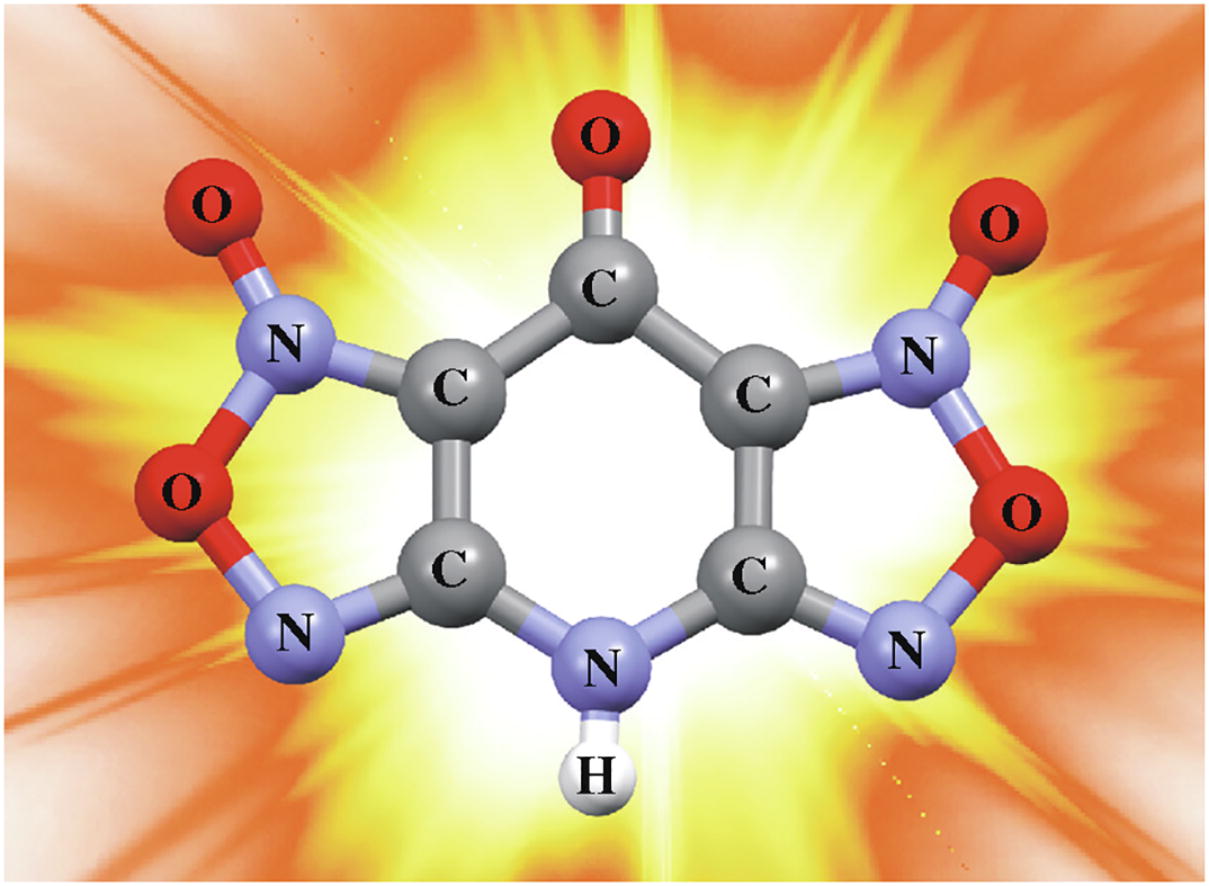
The synthesis and properties of novel high-dense energetic material based on new 4H,8H-bis(furazano)[3,4-b:3',4'-e]-pyridine ring system are described. An operationally simple, efficient and scalable protocol towards 4H,8H-bis(furazano)-[3,4-b:3',4'-e]pyridin-8-one 1,7-dioxide from commercially available 4-amino-2,6-dichloropyridine in three steps, namely, nitration–azidation–cascade ring closure, has been developed.
References
1.

Boyer J.H., McCane D.I., McCarville W.J., Tweedie A.T.
Journal of the American Chemical Society,
1953
2.
Campodónico H.
CEPAL Review,
1999
3.
(a) L. I. Khmel’nitskii, S. S. Novikov and T. I. Godovikova, Khimiya furoksanov: stroenie i sintez (Chemistry of Furoxans: Structure and Synthesis), 2nd edn., Nauka, Moscow, 1996 (in Russian); (b) L. Türker and S. Varis¸, Polycyclic Aromat. Compd., 2009, 29, 228; (c) E. A. Chugunova, A. S. Gazizov, A. R. Burilov, L. M. Yusupova, M. A. Pudovik and O. G. Sinyashin, Russ. Chem. Bull., Int. Ed., 2019, 68, 887 (Izv. Akad. Nauk, Ser. Khim., 2019, 887).
4.

Fershtat L.L., Makhova N.N.
ChemPlusChem,
2019
5.

Zhang J., Zhou J., Bi F., Wang B.
Chinese Chemical Letters,
2020
6.
Zlotin S.G., Churakov A.M., Egorov M.P., Fershtat L.L., Klenov M.S., Kuchurov I.V., Makhova N.N., Smirnov G.A., Tomilov Y.V., Tartakovsky V.A.
Mendeleev Communications,
2021
7.

Zhou J., Zhang J., Wang B., Qiu L., Xu R., Sheremetev A.B.
FirePhysChem,
2022
8.
(a) I. B. Starchenkov and V. G. Andrianov, Chem. Heterocycl. Compd., 1996, 32, 618 (Khim. Geterotsikl. Soedin., 1996, 717); (b) A. B. Sheremetev and I. L. Yudin, Mendeleev Commun., 1996, 6, 247; (c) B. B. Averkiev, A. A. Korlyukov, M. Yu. Antipin, A. B. Sheremetev and T. V. Timofeeva, Cryst. Growth Des., 2014, 14, 5418; (d) D. B. Lempert, A. I. Kazakov, A. B. Sheremetev, A. G. Gladyshkin, A. V. Nabatova and L. S. Yanovskii, Russ. Chem. Bull., Int. Ed., 2019, 68, 1856 (Izv. Akad. Nauk, Ser. Khim., 2019, 1856).
9.
(a) A. A. Astrat’ev, D. V. Dashko and A. I. Stepanov, Cent. Eur. J. Chem., 2012, 10, 1087; (b) S. M. Aldoshin, D. B. Lempert, T. K. Goncharov, A. I. Kazakov, S. I. Soglasnova, E. M. Dorofeenko and N. A. Plishkin, Russ. Chem. Bull., Int. Ed., 2016, 65, 2018 (Izv. Akad. Nauk, Ser. Khim., 2016, 2018); (c) Z. G. Aliev, T. K. Goncharov, D. V. Dashko, E. L. Ignat’eva, A. A. Vasil’eva, N. I. Shishov, D. V. Korchagin, Yu. M. Milekhin and S. M. Aldoshin, Russ. Chem. Bull., Int. Ed., 2017, 66, 694 (Izv. Akad. Nauk, Ser. Khim., 2017, 694); (d) D. B. Lempert, V. V. Raznoschikov and L. S. Yanovskiy, Russ. J. Appl. Chem., 2019, 92, 1690 (Zh. Prikl. Khim., 2019, 92, 1578).
10.
Sheremetev A.B., Kulagina V.O., Yudin I.L., Kuzmina N.E.
Mendeleev Communications,
2001
11.
(b) I. N. Zyuzin, K. Yu. Suponitsky and A. B. Sheremetev, J. Heterocycl. Chem., 2012, 49, 561
12.
![Advances in the chemistry of furazano[3,4-b]pyrazines and their analogues](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Sheremetev A.B., Yudin I.L.
Russian Chemical Reviews,
2003
13.
![Synthesis, characterization and properties of heat-resistant explosive materials: polynitroaromatic substituted difurazano[3,4-b:3′,4′-e]pyrazines](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Liu N., Shu Y., Li H., Zhai L., Li Y., Wang B.
RSC Advances,
2015
14.
![Synthesis of 4,8-Dinitraminodifurazano[3, 4-b,e ]pyrazine Derived Nitrogen-Rich Salts as Potential Energetic Materials](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Li W., Tian J., Qi X., Wang K., Jin Y., Wang B., Zhang Q.
ChemistrySelect,
2018
15.

Li W., Wang K., Qi X., Jin Y., Zhang Q.
Crystal Growth and Design,
2018
16.

Snyder C.J., Wells L.A., Chavez D.E., Imler G.H., Parrish D.A.
Chemical Communications,
2019
17.
(a) A. I. Stepanov, A. A. Astrat’ev, D. V. Dashko, N. P. Spiridonova, S. F. Mel’nikova and I. V. Tselinskii, Russ. Chem. Bull., Int. Ed., 2012, 61, 1024 (Izv. Akad. Nauk, Ser. Khim., 2012, 1019); (b) A. I. Stepanov, D. V. Dashko and A. A. Astrat’ev, Chem. Heterocycl. Compd., 2013, 49, 1068; (c) B.-H. Duan, N. Liu, B. Wang, X. Lu, H. Mo, M. Xu and Y. Shu, Chin. J. Explos. Propellants, 2019, 42, 114, doi: 10.14077/j. issn.1007-7812.2019.02.002; (d) A. I. Kazakov, D. B. Lempert, A. V. Nabatova, D. V. Dashko, V. V. Raznoschikov, L. S. Yanovskii and S. M. Aldoshin, Russ. J. Appl. Chem., 2019, 92, 1696 (Zh. Prikl. Khim., 2019, 92, 1657); (e) L. Zhai, F. Bi, H. Huo, Y. Luo, X. Li, S. Chen and B. Wang, Front. Chem., 2019, 7, doi.org/10.3389/fchem.2019.00559; (f) D. B. Lempert, A. I. Kazakov, A. V. Nabatova, D. V. Dashko, A. I. Stepanov, G. V. Shilov and S. M. Aldoshin, Combust., Explos. Shock Waves, 2020, 56, 621 [Fiz. Goreniya Vzryva, 2020, 56 (6), 3]. (g) N. Liu, C. Xiao, B. Duan, X. Lu, B. Wang, J. Zhang and Q.-L. Yan, ACS Appl. Energy Mater., 2020, 7129.
18.
![Synthesis and thermal behavior of a fused, tricyclic pyridine-based energetic material: 4-amino-5-nitro-[1,2,5]oxadiazolo[3,4-e]tetra-zolo[1,5-a]pyridine-3-oxide](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ma C., Pan Y., Jiang J., Liu Z., Yao Q.
New Journal of Chemistry,
2018
19.

Wilkinson J.H., Finar I.L.
Journal of the Chemical Society (Resumed),
1948
20.

Tatibouët A., Demeunynck M., Lhomme J.
Synthetic Communications,
1996
21.

Hampton A., Magrath D.
Journal of the Chemical Society (Resumed),
1949
22.
(b) K. H. Oh, J. G. Kim and J. K. Park, Org. Lett., 2017, 19, 3994.
23.

Dalinger I.L., Suponitsky K.Y., Shkineva T.K., Lempert D.B., Sheremetev A.B.
Journal of Materials Chemistry A,
2018
24.

Palysaeva N.V., Gladyshkin A.G., Vatsadze I.A., Suponitsky K.Y., Dmitriev D.E., Sheremetev A.B.
Organic Chemistry Frontiers,
2019
25.

Suponitsky K.Y., Smol'yakov A.F., Ananyev I.V., Khakhalev A.V., Gidaspov A.A., Sheremetev A.B.
ChemistrySelect,
2020
26.

Sirach R.R., Dave P.N.
Chemistry of Heterocyclic Compounds,
2021
27.
Fizika Vzryva (Physics of Explosion), ed. L. P. Orlenko, 3rd edn., Fizmatlit, Moscow, 2002, vol. 1 (in Russian).