Keywords
antimony
crystal structure
iodine
non-covalent interactions
organoantimony compounds
organometallic compounds
Abstract
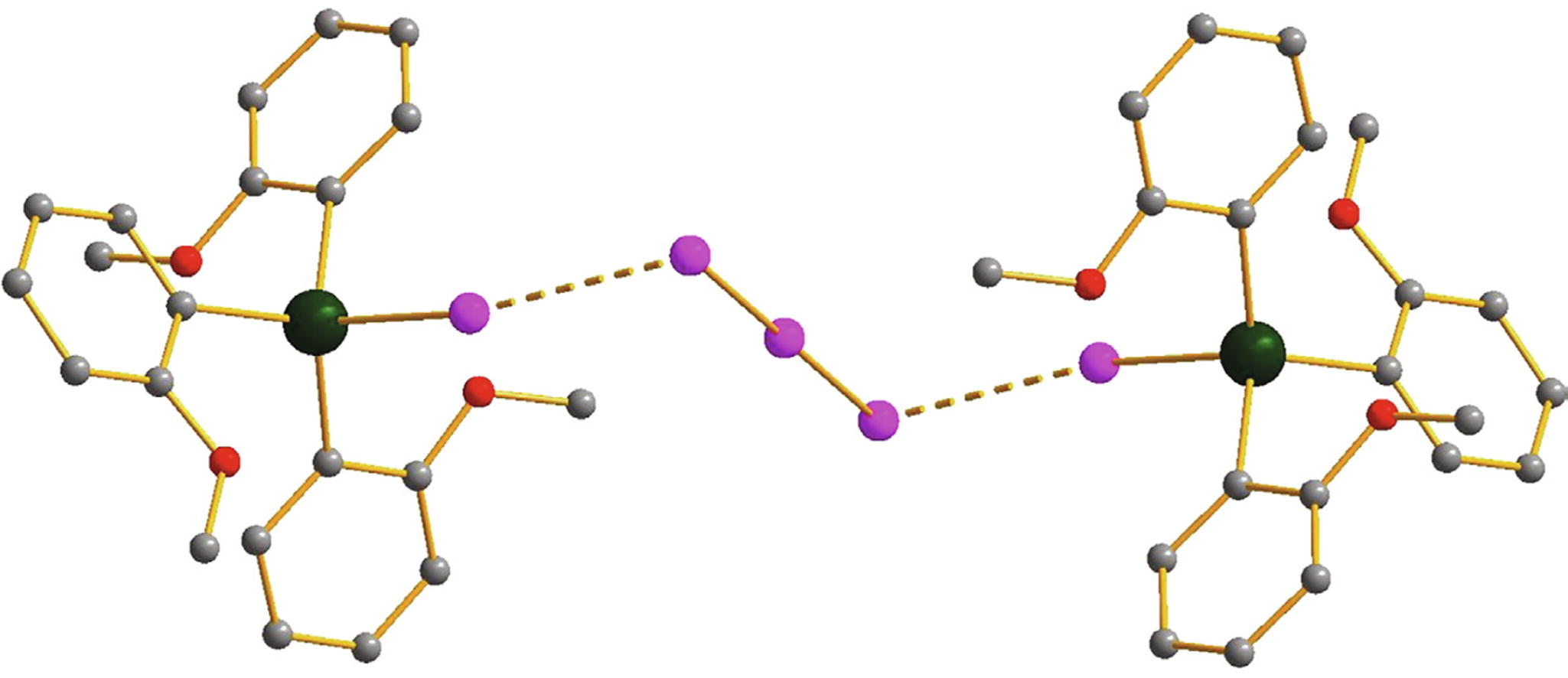
The reaction of 2-methoxyphenyllithium with SbCl3 affords tris(2-methoxyphenyl)antimony. Its treatment with equimolar amount of I2 results in stiborane (2-MeOC6H4)3SbI2, while heating with 2 equivalents of I2 leads to iodotris(2-methoxyphenyl)stibonium triiodide [(2-MeOC6H4)3SbI]+ [I3]– featuring halogen bonding in solid state.
References
1.

Sharutin V.V., Sharutina O.K., Efremov A.N.
Russian Journal of Inorganic Chemistry,
2020
2.

Bukhvalova S.Y., Zhiganshina E.R., Astaf’eva T.V., Arsenyev M.V., Baranov E.V., Chesnokov S.A., Poddel’sky A.I.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2020
3.

Sharutin V.V., Poddel’sky A.I., Sharutina O.K.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2020
4.
L. S. Okhlopkova, A. I. Poddel’sky, I. V. Smolyaninov and G. K. Fukin, Russ. J. Coord. Chem., 2020, 46, 386 (Zh. Koord. Khim., 2020, 46, 340).
5.

Sharutin V.V., Sharutina O.K.
Russian Journal of General Chemistry,
2016
6.
![Recognition of linear and bent forms of solid μ-oxo-bis[iodotriphenylantimony(V)] (Ph3SbI)2O](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Taylor M.J., Baker L.-., Rickard C.E., Surman P.W.
Journal of Organometallic Chemistry,
1995
7.
![Substituent-dependent reactivity of triarylantimony(iii) toward I2: isolation of [Ar3SbI]+ salt](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Sharutin V.V., Sharutina O.K., Novikov A.S., Adonin S.A.
New Journal of Chemistry,
2020
8.
](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Torubaev Y.V., Skabitsky I.V.
Zeitschrift fur Kristallographie - Crystalline Materials,
2020
9.

Torubaev Y.V., Lyssenko K.A., Popova A.E.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2019
10.

Ivanov D.M., Kinzhalov M.A., Novikov A.S., Ananyev I.V., Romanova A.A., Boyarskiy V.P., Haukka M., Kukushkin V.Y.
Crystal Growth and Design,
2017
11.

Eliseeva A.A., Ivanov D.M., Novikov A.S., Rozhkov A.V., Kornyakov I.V., Dubovtsev A.Y., Kukushkin V.Y.
Dalton Transactions,
2020
12.

Soldatova N.S., Suslonov V.V., Kissler T.Y., Ivanov D.M., Novikov A.S., Yusubov M.S., Postnikov P.S., Kukushkin V.Y.
Crystals,
2020
13.

Eliseeva A.A., Ivanov D.M., Novikov A.S., Kukushkin V.Y.
CrystEngComm,
2019
14.

Wu J., Hu Z., Sung H.
Journal of Molecular Structure,
2021
15.

Sivchik V.V., Solomatina A.I., Chen Y., Karttunen A.J., Tunik S.P., Chou P., Koshevoy I.O.
Angewandte Chemie - International Edition,
2015
16.

Bulatova M., Melekhova A.A., Novikov A.S., Ivanov D.M., Bokach N.A.
Zeitschrift fur Kristallographie - Crystalline Materials,
2018
17.

Bondi A.
The Journal of Physical Chemistry,
1966
18.

Mantina M., Chamberlin A.C., Valero R., Cramer C.J., Truhlar D.G.
Journal of Physical Chemistry A,
2009