Keywords
1,2,3-triazoles
calix[4]resorcinols
click reaction
condensation
dendrimers
dendrons
Abstract
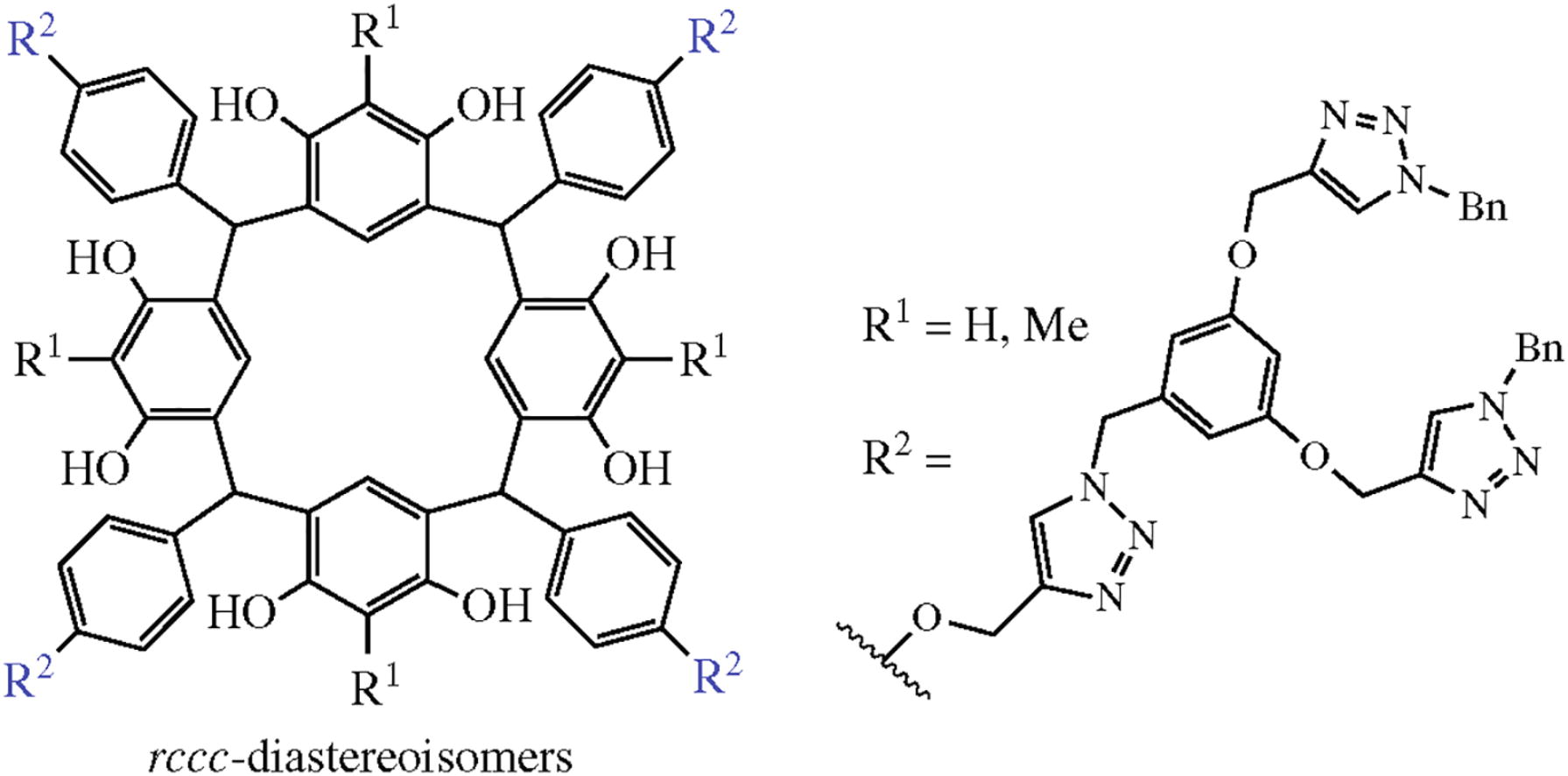
Novel first-generation dendrimers on the calix[4]resorcinol core with four branches each containing multiple 1,2,3-triazole units have been synthesized in one-step by acid catalyzed condensation of resorcinols with a new aldehyde dendron, namely, 4-{3,5-bis[(1-benzyl-1H-1,2,3-triazol-4-yl)- methoxy]benzyloxy}benzaldehyde (obtained by alkyne–azide cycloaddition). The reaction proceeds stereoselectively to form rccc-diastereoisomers in high yields.
References
1.

Beletskaya I.P., Chuchurjukin A.V.
Russian Chemical Reviews,
2000
2.
Klajnert B., Bryszewska M.
Acta Biochimica Polonica,
2001
3.

Grayson S.M., Fréchet J.M.
Chemical Reviews,
2001
4.

Astruc D., Chardac F.
Chemical Reviews,
2001
5.

Caminade A., Laurent R., Zablocka M., Majoral J.
Molecules,
2012
6.

Rasmussen B., Christensen J.B.
Organic and Biomolecular Chemistry,
2012
7.

Mignani S., Shi X., Ceña V., Shcharbin D., Bryszewska M., Majoral J.
Drug Discovery Today,
2021
8.

Shiri P., Amani A.M.
Monatshefte fur Chemie,
2021
9.
![A New Photoresist Based on Calix[4]resorcinarene Dendrimer](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Haba O., Haga K., Ueda M., Morikawa O., Konishi H.
Chemistry of Materials,
1999
10.
![Amphiphilicp-tert-Butylcalix[4]arene Scaffolds Containing Exposed Carbohydrate Dendrons](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Roy R., Kim J.M.
Angewandte Chemie - International Edition,
1999
11.
![Rapid synthesis of dendrimers based on calix[4]resorcinarenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Yamakawa Y., Ueda M., Nagahata R., Takeuchi K., Asai M.
Journal of the Chemical Society Perkin Transactions 1,
1998
12.
![Lysine dendrimers based on thiacalix[4]arene core moieties as molecular scaffolds for supramolecular host systems](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Appelhans D., Smet M., Khimich G., Komber H., Voigt D., Lhoták P., Kuckling D., Voit B.
New Journal of Chemistry,
2005
13.
![Syntheses and metal ions recognition of dendritic calix[n]arenes (n = 6,8) amide derivative](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Wang Y., Cai Y., Yan C.
Frontiers of Chemistry in China,
2007
14.

Marradi M., Cicchi S., Sansone F., Casnati A., Goti A.
Beilstein Journal of Organic Chemistry,
2012
15.
![Synthesis, Recognition of Metal Ions of Salicylidenimine Functionalized p-tert-Butylcalix[n]arene-core Dendrimers](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Wang Y., Cai Y., Yan C.
Supramolecular Chemistry,
2007
16.
![Calixarene-based dendrimers. Second generation of a calix[4]-dendrimer with a ‘tren’ as core](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Cheriaa N., Abidi R., Vicens J.
Tetrahedron Letters,
2005
17.

Mahouachi M., Kim Y., Lee S.H., Abidi R., Harrowfield J., Vicens J.
Supramolecular Chemistry,
2005
18.

Xu H., Kinsel G.R., Zhang J., Li M., Rudkevich D.M.
Tetrahedron,
2003
19.

Li H., Sun J., Cullum B.M.
Nanobiotechnology,
2006
20.

Lambert J.B., Kang S., Ma K., Liu C., Condie A.G.
Journal of Organic Chemistry,
2009
21.
10.1016/j.mencom.2022.01.033_b0105
Cheriaa
J. Nanobiotechnol.,
2005
22.

Baklouti L., Cheriaa N., Mahouachi M., Abidi R., Kim J.S., Kim Y., Vicens J.
Journal of Inclusion Phenomena and Macrocyclic Chemistry,
2006
23.

Kim J.S., Lee S.Y., Yoon J., Vicens J.
Chemical Communications,
2009
24.

Lalor R., Gunning A.P., Morris V.J., Matthews S.E.
Chemical Communications,
2010
25.

Lijanova I.V., Moggio I., Arias E., Vazquez-Garcia R., Martínez-García M.
Journal of Nanoscience and Nanotechnology,
2007
26.

Lijanova I.V., Moggio I., Arias E., Klimova T., Martínez-García M.
Tetrahedron,
2008
27.

Victorovna-Lijanova I., Reyes-Valderrama M.I., Maldonado J., Ramos-Ortiz G., Tatiana K., Martínez-García M.
Tetrahedron,
2008
28.
![Synthesis of Calix[4] resorcinarene Core Dendrimers with Peripheral Salicylideneimine Functional Groups](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
HAN J., GE Y., YAN C.
Chemical Research in Chinese Universities,
2006
29.

Dominguez-Chávez J.G., Lijanova I., Moggio I., Arias E., Vazquez-Garcia R.A., Ma I.R., Klimova T., Gutierrez-Nava M., Martínez-García M.
Journal of Nanoscience and Nanotechnology,
2007
30.

Lijanova I.V., Berestneva T.K., García M.M.
Supramolecular Chemistry,
2007
31.

Wu P., Feldman A.K., Nugent A.K., Hawker C.J., Scheel A., Voit B., Pyun J., Fréchet J.M., Sharpless K.B., Fokin V.V.
Angewandte Chemie - International Edition,
2004
32.
![One-step synthesis of rccc- and rctt-diastereomers of novel calix[4]resorcinols based on a para-thiophosphorylated derivative of benzaldehyde](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva I.R., Sokolova V.I., Gruner M., Habicher W.D., Syakaev V.V., Khrizanforova V.V., Gabidullin B.M., Gubaidullin A.T., Budnikova Y.H., Burilov A.R., Pudovik M.A.
Tetrahedron Letters,
2013
33.
![Synthesis of rccc- and rctt-diastereoisomers of novel triazole-containing calix[4]resorcinols](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva I.R., Mukhamedyanova K.M., Syakaev V.V., Gubaidullin A.T., Habicher W.D., Burilov A.R.
Tetrahedron Letters,
2018
34.
Knyazeva I.R., Syakaev V.V., Lodochnikova O.A., Burilov A.R.
Mendeleev Communications,
2019
35.
Knyazeva I.R., Gerasimova T.P., Kolesnikov I.E., Syakaev V.V., Katsyuba S.A., Burilov A.R.
Mendeleev Communications,
2020