Keywords
acetaldehyde
acetals
Diels–Alder adducts
levoglucosenone
McMurry reaction
pinacol reaction
Abstract
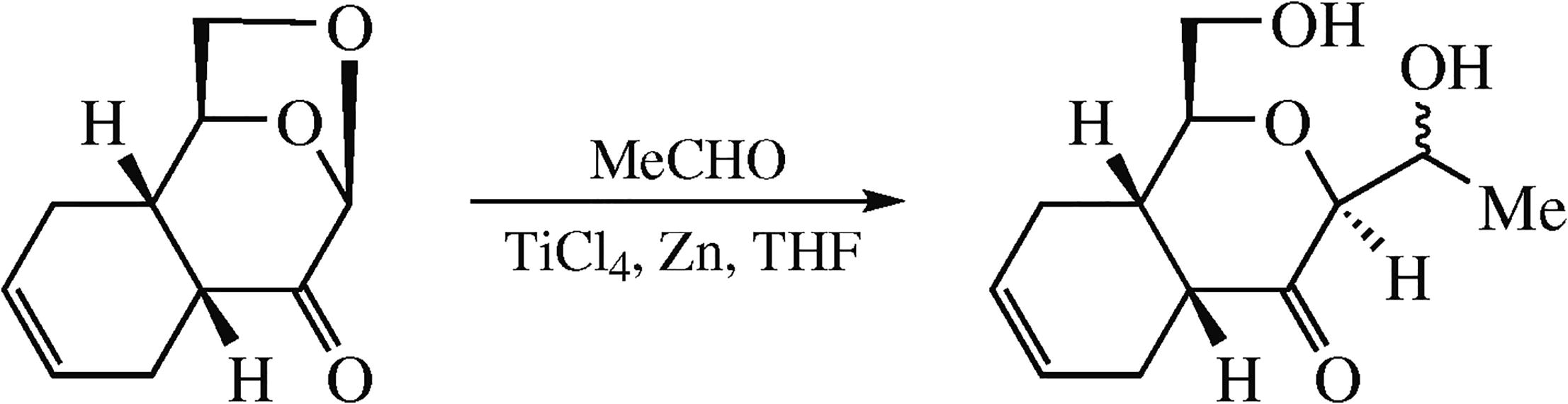
Reactions of acetaldehyde with Diels–Alder adducts of levoglucosenone with butadiene, isoprene, and cyclohexa-diene assisted by low-valence titanium afford products of acetaldehyde addition to the acetal center with opening of the 1,6-anhydro bridge. In the case of the cyclopentadiene adduct, the reaction gives the product of addition of the ethyl substituent to the acetal center while the 1,6-anhydro bridge remains unchanged.
References
1.
(a) M. Isobe, N. Fukami, T. Nishikava and T. Goto, Heterocycles, 1987, 25, 521; (b) P. Bhate and D. Horton, Carbohydr. Res., 1983, 122, 189; (c) A.M. Sarotti, R. A. Spanevello, C. Duhayon, J.-P. Tuchagues and A. G. Suárez, Tetrahedron, 2007, 63, 241; (d) D. D. Ward and F. Shafizadeh, Carbohydr. Res., 1981, 95, 155; (e) G. A. Tolstikov, F. A. Valeev, I. N. Gaisina, L. V. Spirikhin and M. S. Miftakhov, Russ. J. Org. Chem., 1992, 28, 1659 (Zh. Org. Khim., 1992, 28, 2072); (f) F. A. Valeev, I. N. Gaisina and M. S. Miftakhov, Russ. Chem. Bull., 1996, 45, 2453 (Izv. Akad. Nauk, Ser. Khim., 1996, 2584); (g) M. S. Miftakhov, I. N. Gaisina and F. A. Valeev, Russ. Chem. Bull., 1996, 45, 1942 (Izv. Akad. Nauk, Ser. Khim., 1996, 2047); (h) A. M. Sarotti, A. G. Suárez and R. A. Spanevello, Tetrahedron Lett., 2011, 52, 3116; (i) M. S. Miftakhov, F. A. Valeev and I. N. Gaisina, Russ. Chem. Rev., 1994, 63, 869 (Usp. Khim., 1994, 63, 922); (j) Levoglucosenone and Levoglucosans: Chemistry and Applications, ed. Z. J. Witczak, ATL Press, Mount Prospect, IL, 1994; (k) M. S. Miftakhov, F. A. Valeev, I. N. Gaisina and G. A. Tolstikov, Mendeleev Commun., 1994, 4, 45; (l) M. Bamba, T. Nishikawa and M. Isobe, Tetrahedron, 1998, 54, 6639; (m) A. M. Sarotti, M. Zanardi and R. A. Spanevello, Curr. Org. Synth., 2012, 9, 439; (n) M. B. Comba, Y. Tsai, A. M. Sarotti, M. I. Mangione, A. G. Suárez and R. A. Spanevello, Eur. J. Org. Chem., 2018, 590; (o) L. Kh. Faizullina, Yu. A. Khalilova and F. A. Valeev, Mendeleev Commun., 2021, 31, 493.
2.

Tagirov A.R., Biktagirov I.M., Galimova Y.S., Faizullina L.K., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2015
3.
A. R. Tagirov, Yu. S. Galimova, L. Kh. Faizullina, L. V. Spirikhin, Sh. M. Salikhov and F. A. Valeev, Russ. J. Org. Chem., 2017, 53, 105 (Zh. Org. Khim., 2017, 53, 1040).
4.

Biktagirov I.M., Faizullina L.K., Salikhov S.M., Iskakova M.M., Safarov M.G., Galin F.Z., Valeev F.A.
Russian Journal of Organic Chemistry,
2015
5.

McMurry J.E., Fleming M.P., Kees K.L., Krepski L.R.
Journal of Organic Chemistry,
1978
6.

McMurry J.E., Farina V.
Tetrahedron Letters,
1983
7.

Corey E.J., Danheiser R.L., Chandrasekaran S.
Journal of Organic Chemistry,
1976
8.

McMurry J.E.
Chemical Reviews,
1989
9.

McMurry J.E., Krepski L.R.
Journal of Organic Chemistry,
1976
10.

Coe P.L., Scriven C.E.
Journal of the Chemical Society Perkin Transactions 1,
1986
11.

Biktagirov I.M., Faizullina L.K., Salikhov S.M., Galin F.Z., Safarov M.G., Valeev F.A.
Russian Journal of Organic Chemistry,
2016
12.

Mukaiyama T., Narasaka K., Banno K.
Chemistry Letters,
1973
13.

Balme G., Gore J.
Journal of Organic Chemistry,
1983