Keywords
19F NMR
absolute configuration
alkaloids
Grignard reaction
organofluorine compounds
orvinols
thevinols
thevinone
Abstract
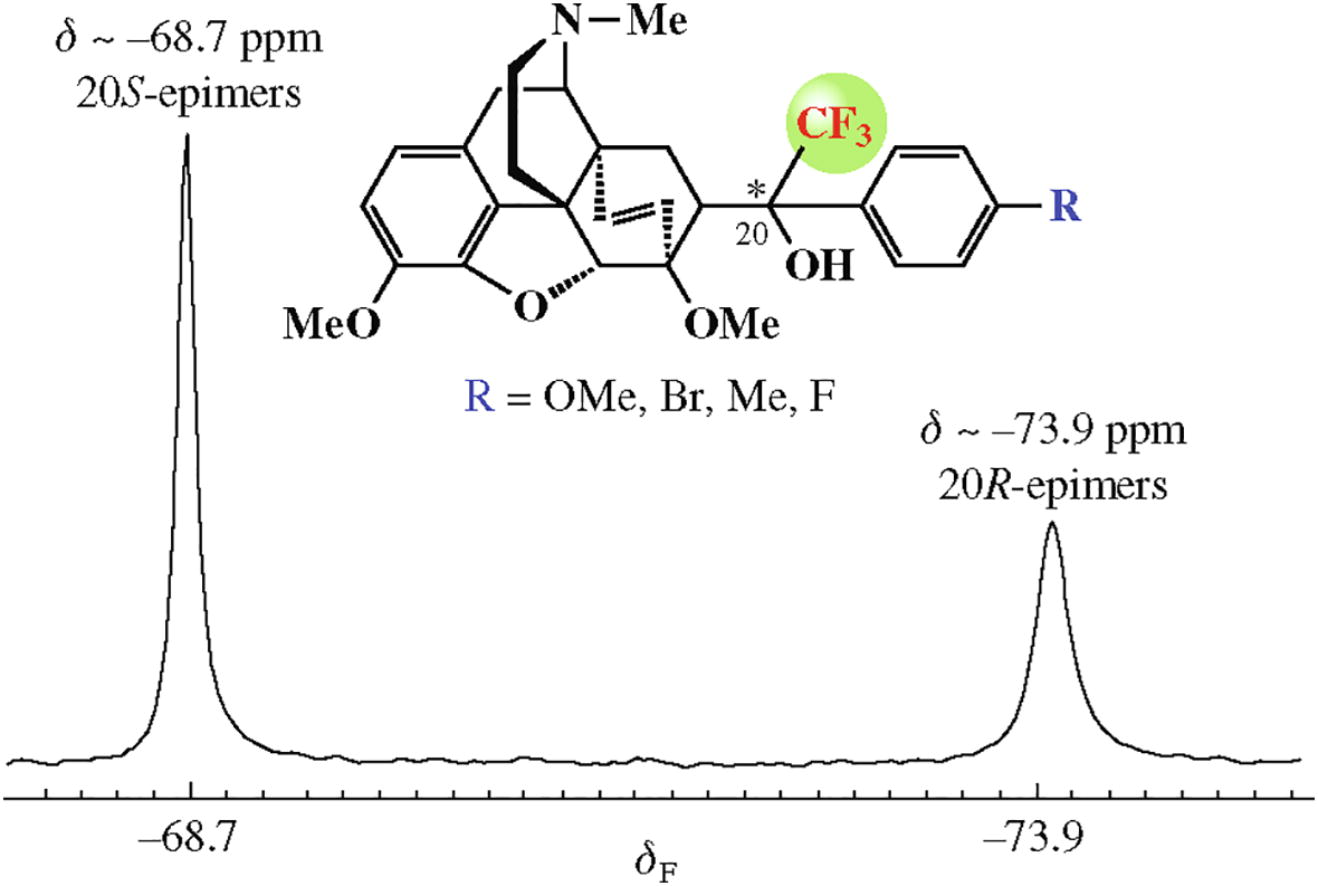
21,21,21-Trifluoro(aryl)thevinols were synthesized by the reaction of 21,21,21-trifluorothevinone with aryl Grignard reagents. The absolute configuration at C20 atom in these CF3-substituted alcohols can be easily determined by one-dimensional 19F NMR spectroscopy that was verified by X-ray diffraction studies.
References
1.

Bentley K.W., Hardy D.G.
Journal of the American Chemical Society,
1967
2.
10.1016/j.mencom.2022.01.031_b0010
Lenz
Opiates,
1986
3.
10.1016/j.mencom.2022.01.031_b0015
Opioid Analgesics: Chemistry and Receptors,
1986
4.

Lewis J.W., Husbands S.M.
Topics in Current Chemistry,
2010
5.

Politanskaya L.V., Selivanova G.A., Panteleeva E.V., Tretyakov E.V., Platonov V.E., Nikul’shin P.V., Vinogradov A.S., Zonov Y.V., Karpov V.M., Mezhenkova T.V., Vasilyev A.V., Koldobskii A.B., Shilova O.S., Morozova S.M., Burgart Y.V., et. al.
Russian Chemical Reviews,
2019
6.

Purser S., Moore P.R., Swallow S., Gouverneur V.
Chemical Society Reviews,
2008
7.
![Fluorinated and [18F]fluorinated morphinan based opioid ligands](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Sandulenko I.V., Ambartsumyan A.A., Moiseev S.K.
Organic and Biomolecular Chemistry,
2020
8.

Sandulenko I.V., Kovaleva E.S., Peregudov A.S., Kalinin V.N., Moiseev S.K.
ChemistrySelect,
2016
9.

Bentley K.W., Hardy D.G., Meek B.
Journal of the American Chemical Society,
1967
10.
10.1016/j.mencom.2022.01.031_b0050
Burger’s Medicinal Chemistry,
1982
11.

Lewis J., Husbands S.
Current Pharmaceutical Design,
2004
12.

Hutchins C.W., Cooper G.K., Puerro S., Rapoport H.
Journal of Medicinal Chemistry,
1981
13.

Batta G., Gunda T.E., Szabó Z., Berényi S., Gulyás G., Makleit S.
Magnetic Resonance in Chemistry,
1992
14.

Dale J.A., Mosher H.S.
Journal of the American Chemical Society,
1973
15.

Ohtani I., Kusumi T., Kashman Y., Kakisawa H.
Journal of the American Chemical Society,
1991
16.

Rieser M.J., Hui Y.H., Rupprecht J.K., Kozlowski J.F., Wood K.V., McLaughlin J.L., Hanson P.R., Zhuang Z., Hoye T.R.
Journal of the American Chemical Society,
1992
17.