Keywords
amination
copper
heterogeneous catalysis
homogeneous catalysis
iodoarenes
nanoparticles
Abstract
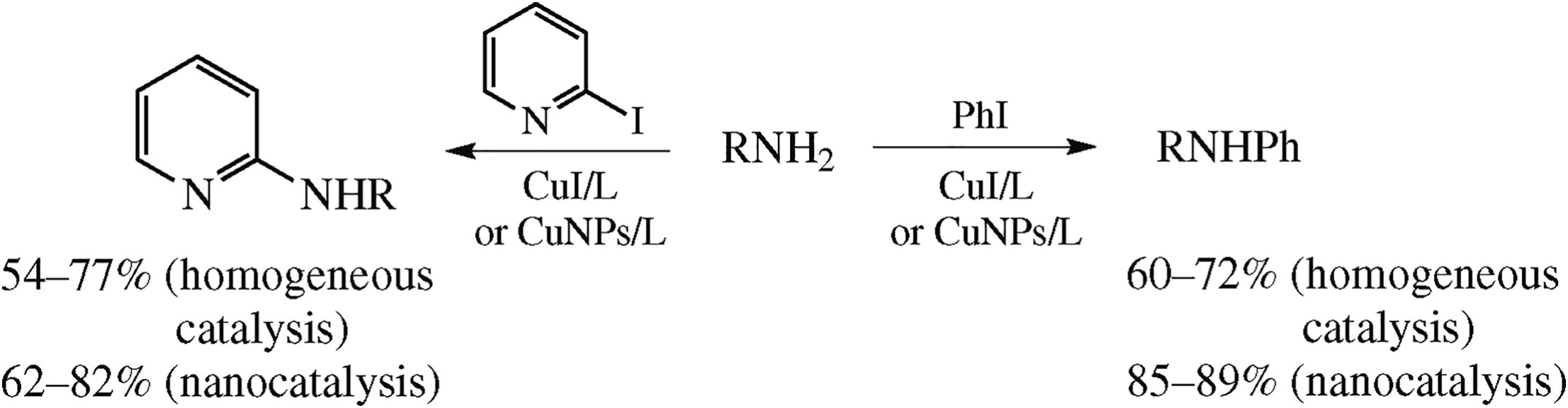
Comparison of the homogeneous and heterogeneous copper-catalyzed arylation of model primary amines with (hetero)-aryl iodides in DMSO revealed a comparable efficiency of CuI and commercially available unsupported copper nanoparticles (25 nm size) in the presence of 2-isobutyryl-cyclohexanone or L-proline.
References
1.

Ingoglia B.T., Wagen C.C., Buchwald S.L.
Tetrahedron,
2019
2.

Sambiagio C., Marsden S.P., Blacker A.J., McGowan P.C.
Chemical Society Reviews,
2014
3.

Okano K., Tokuyama H., Fukuyama T.
Chemical Communications,
2014
4.

Neetha M., Saranya S., Ann Harry N., Anilkumar G.
ChemistrySelect,
2020
5.
10.1016/j.mencom.2022.01.029_b0025
Shaughnessy
Copper-Catalyzed Amination of Aryl and Alkenyl Electrophiles,
2017
6.
10.1016/j.mencom.2022.01.029_b0030
Tang
Synthesis,
2008
7.

Rout L., Jammi S., Punniyamurthy T.
Organic Letters,
2007
8.

Thathagar M.B., Beckers J., Rothenberg G.
Journal of the American Chemical Society,
2002
9.

Thathagar M.B., Beckers J., Rothenberg G.
Green Chemistry,
2004
10.

Beletskaya I., Abel A., Averin A., Buryak A., Savelyev E., Orlinson B., Novakov I.
Synthesis,
2017
11.

Averin A.D., Panchenko S.P., Abel A.S., Maloshitskaya O.A., Butov G.M., Savelyev E.N., Orlinson B.S., Novakov I.A., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2017
12.

Baxter A., Bent J., Bowers K., Braddock M., Brough S., Fagura M., Lawson M., McInally T., Mortimore M., Robertson M., Weaver R., Webborn P.
Bioorganic and Medicinal Chemistry Letters,
2003
13.

Banie H., Sinha A., Thomas R.J., Sircar J.C., Richards M.L.
Journal of Medicinal Chemistry,
2007
14.

Tice C.M., Zhao W., Krosky P.M., Kruk B.A., Berbaum J., Johnson J.A., Bukhtiyarov Y., Panemangalore R., Scott B.B., Zhao Y., Bruno J.G., Howard L., Togias J., Ye Y., Singh S.B., et. al.
Bioorganic and Medicinal Chemistry Letters,
2010
15.

Lyakhovich M.S., Murashkina A.V., Averin A.D., Abel A.S., Maloshitskaya O.A., Savelyev E.N., Orlinson B.S., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2019
16.

Lyakhovich M.S., Murashkina A.V., Panchenko S.P., Averin A.D., Abel A.S., Maloshitskaya O.A., Savelyev E.N., Orlinson B.S., Novakov I.A., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2021
17.
Abel A.S., Kharlamova A.D., Averin A.D., Beletskaya I.P.
Mendeleev Communications,
2021
18.

Averin A.D., Abel A.S., Grigorova O.K., Latyshev G.V., Kotovshchikov Y.N., Mitrofanov A.Y., Bessmertnykh-Lemeune A., Beletskaya I.P.
Pure and Applied Chemistry,
2020
19.

SHILIMKAR T., ANUSE M.
Separation and Purification Technology,
2002
20.

Shilimkar T.N., Kolekar S.S., Anuse M.A.
Separation and Purification Technology,
2005
21.

Mane C.P., Mahamuni S.V., Kolekar S.S., Han S.H., Anuse M.A.
Arabian Journal of Chemistry,
2016
22.

23.

Lokhande T.
Talanta,
1998
24.

Suryavanshi V.J., Patil M.M., Zanje S.B., Kokare A.N., Kore G.D., Anuse M.A., Mulik G.N.
Separation Science and Technology,
2016
25.

Suryavanshi V.J., Patil M.M., Zanje S.B., Kokare A.N., Gaikwad A.P., Anuse M.A., Mulik G.N.
Russian Journal of Inorganic Chemistry,
2017
26.

Kore G.D., Zanje S.B., Kokare A.N., Suryavanshi V.J., Anuse M.A., Kolekar S.S.
Journal of Radioanalytical and Nuclear Chemistry,
2021
27.
10.1016/j.mencom.2022.01.029_b0135
Wahyudi
Sci. Eng.,
2018
28.

Mondal P., Sinha A., Salam N., Roy A.S., Jana N.R., Islam S.M.
RSC Advances,
2013
29.

Gawande M.B., Goswami A., Felpin F., Asefa T., Huang X., Silva R., Zou X., Zboril R., Varma R.S.
Chemical Reviews,
2016
30.

Mitrofanov A.Y., Murashkina A.V., Martín-García I., Alonso F., Beletskaya I.P.
Catalysis Science and Technology,
2017
31.
10.1016/j.mencom.2022.01.029_b0155
Esmaeilpour
Appl. Organomet. Chem.,
2018