Keywords
4-(trimethylsilyl)phenyl group
fluorescence
fluorescence quantum yield
organosilicon compounds
rotation
Suzuki–Miyaura coupling
transition moment
triphenylene
Abstract
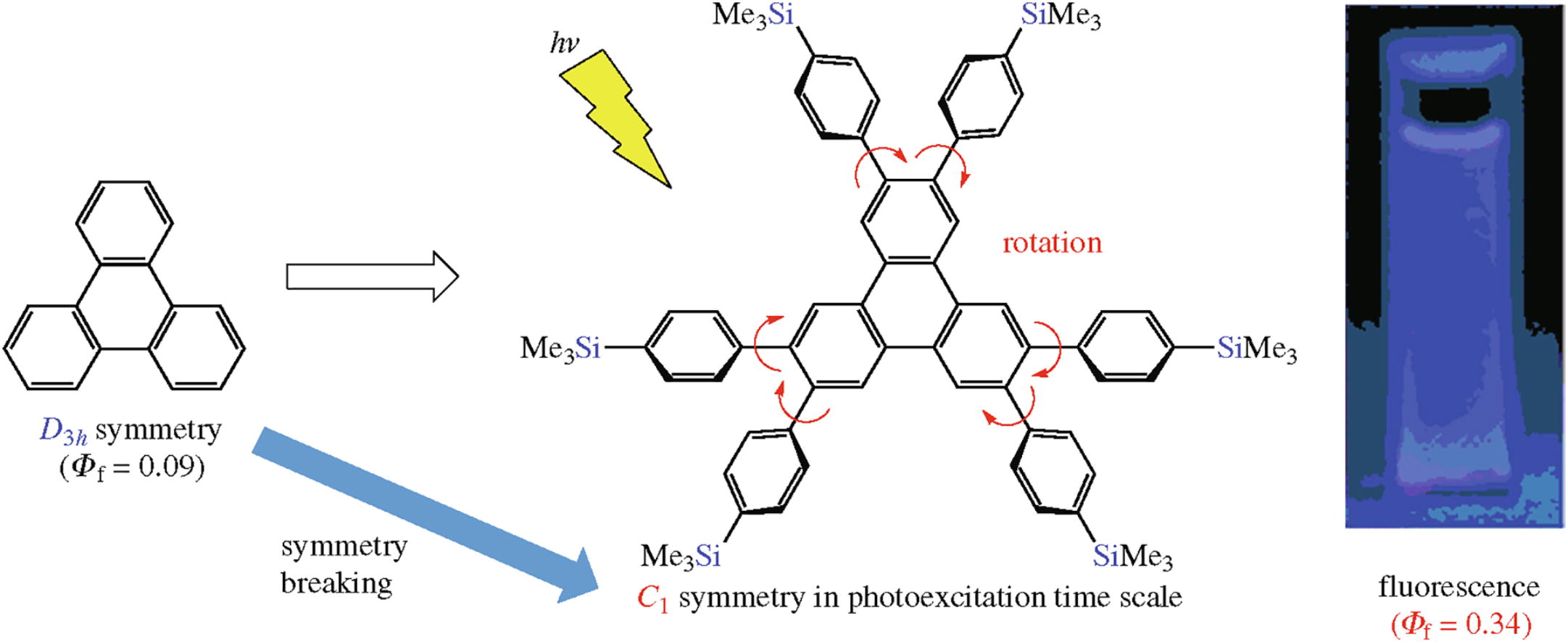
The fluorescence quantum yield of triphenylene is low because the transition moment between the HOMOs and the LUMOs is negligibly small. To improve the fluorescence quantum yield, break of the symmetrical structure of triphenylene is necessary. We found that the rotational effect of the 4-(trimethylsilyl)phenyl groups of 2,3,6,7,10,11-hexakis[4-(trimethylsilyl)phenyl]triphenylene is effective to break the symmetry in spite of the symmetrical structure, leading to improvement of the fluorescence quantum yield.
References
1.

Tang C.W., VanSlyke S.A.
Applied Physics Letters,
1987
2.

Burroughes J.H., Bradley D.D., Brown A.R., Marks R.N., Mackay K., Friend R.H., Burns P.L., Holmes A.B.
Nature,
1990
3.

Kido J., Kimura M., Nagai K.
Science,
1995
4.

Chan C., Tanaka M., Lee Y., Wong Y., Nakanotani H., Hatakeyama T., Adachi C.
Nature Photonics,
2021
5.
10.1016/j.mencom.2022.01.028_b0025
Chandrasekhar
Liquid Crystals,
1992
6.
10.1016/j.mencom.2022.01.028_b0030
Handbook of Liquid Crystals,
2014
7.
10.1016/j.mencom.2022.01.028_b0035
Kumar
Chemistry of Discotic Liquid Crystals: From Monomers to Polymers,
2011
8.

Adam D., Closs F., Frey T., Funhoff D., Haarer D., Schuhmacher P., Siemensmeyer K.
Physical Review Letters,
1993
9.

Adam D., Schuhmacher P., Simmerer J., Häussling L., Siemensmeyer K., Etzbachi K.H., Ringsdorf H., Haarer D.
Nature,
1994
10.

Christ T., Greiner A., Sander R., Stümpflen V., Wendorff J.H.
Advanced Materials,
1997
11.

Christ T., Geffart F., Glüsen B., Kettner A., Lüssem G., Schäfer O., Stümpflen V., Wendorff J.H., Tsukruk V.V.
Thin Solid Films,
1997
12.

Christ T., Stümpflen V., Wendorff J.H.
Macromolecular Rapid Communications,
1997
13.

TAPFF I.H., STUMPFLEN V., WENDORFF J.H., SPOHN D.B., MOBIUS D.
Liquid Crystals,
1997
14.

Mori H., Itoh Y., Nishiura Y., Nakamura T., Yukio Shinagawa Y.S.
Japanese Journal of Applied Physics, Part 1: Regular Papers & Short Notes,
1997
15.
10.1016/j.mencom.2022.01.028_b0080
Murov
Handbook of Photochemistry,
1993
16.

Parker C.A., Joyce T.A.
Transactions of the Faraday Society,
1966
17.

Kyushin S., Takemasa N., Matsumoto H., Horiuchi H., Hiratsuka H.
Chemistry Letters,
2003
18.
10.1016/j.mencom.2022.01.028_b0095
Roussel
Tetrahedron Lett.,
1861
19.

Roussel C., Liden A., Chanon M., Metzger J., Sandstrom J.
Journal of the American Chemical Society,
1976
20.

Liden A., Roussel C., Liljefors T., Chanon M., Carter R.E., Metzger J., Sandstrom J.
Journal of the American Chemical Society,
1976
21.

Berg U., Pettersson I.
Magnetic Resonance in Chemistry,
1985
22.

Franck J., Dymond E.G.
Transactions of the Faraday Society,
1926
23.

Condon E.
Physical Review A,
1926
24.

Miyaura N., Yamada K., Suzuki A.
Tetrahedron Letters,
1979
25.
10.1016/j.mencom.2022.01.028_b0130
Miyaura
J. Chem. Soc., Chem. Commun.,
1979
26.

Karatsu T.
Journal of Photochemistry and Photobiology C: Photochemistry Reviews,
2008
27.
![Benzo[1,2:4,5]bis(1,1,2,2-tetraisopropyldisilacyclobutene)](/storage/images/resized/yNSijlgQghQF53VZuyFLA30CKDe4j3HK74Vtpnxa_small_thumb.webp)
Kyushin S., Kitahara T., Matsumoto H.
Chemistry Letters,
1998
28.

Kyushin S., Matsuura T., Matsumoto H.
Organometallics,
2006
29.

Tsurusaki A., Kobayashi A., Kyushin S.
Asian Journal of Organic Chemistry,
2017
30.

Haruki R., Sasaki Y., Masutani K., Yanai N., Kimizuka N.
Chemical Communications,
2020
31.

Kyushin S., Ikarugi M., Goto M., Hiratsuka H., Matsumoto H.
Organometallics,
1996
32.

Kyushin S., Ishikita Y., Matsumoto H., Horiuchi H., Hiratsuka H.
Chemistry Letters,
2006