Keywords
alkenes
amines
esters
ethers
organoselenium compounds
selenation
selenenyl iodides
Abstract
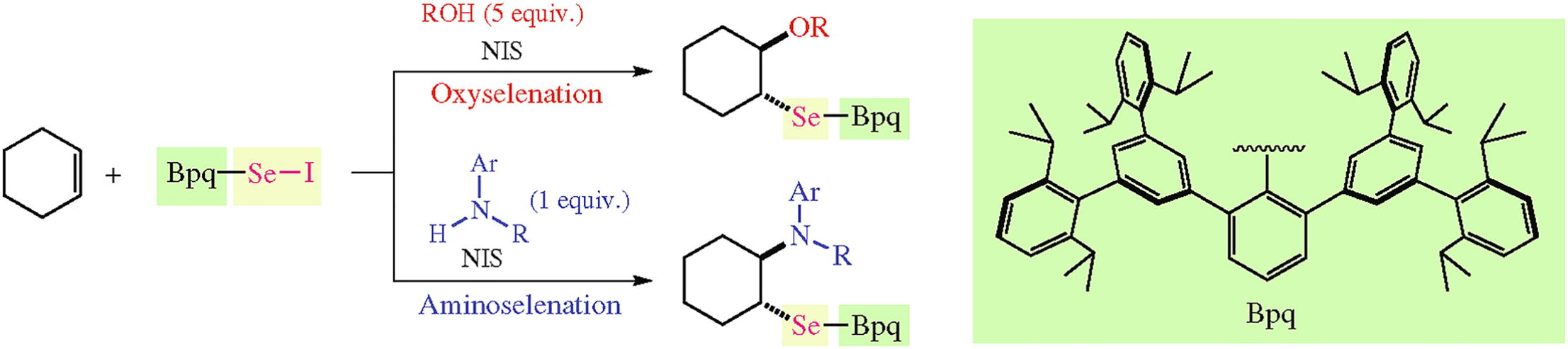
Application of an isolable selenenyl iodide BpqSeI (Bpq denotes 5',5'''-bis(2,6-diisopropylphenyl)-2,6,2'''',6''''-tetra-isopropyl-1,1':3',1'':3'',1''':3''',1''''-quinquephenyl-2''-yl) to selenofunctionalization of alkenes with external oxygen or nitrogen nucleophiles was investigated. In the presence of N-iodosuccinimide as an additive, alcohols, a carboxylic acid, or aromatic amines were smoothly introduced to alkenes to give the corresponding β-oxy or β-amino selenides in moderate to high yields.
References
1.

Reich H.J.
Accounts of Chemical Research,
1979
2.
10.1016/j.mencom.2022.01.026_b0010
Nicolaou
in Selenium in Natural Products Synthesis,
1984
3.

Wirth T.
Angewandte Chemie - International Edition,
2000
4.
10.1016/j.mencom.2022.01.026_b0020
Organoselenium Chemistry: Modern Developments in Organic Synthesis,
2000
5.

Freudendahl D.M., Shahzad S.A., Wirth T.
European Journal of Organic Chemistry,
2009
6.

Campos M.D., Petragnani N.
Chemische Berichte,
1960
7.

Sharpless K.B., Lauer R.F.
Journal of Organic Chemistry,
1974
8.

Nicolaou K.C., Lysenko Z.
Journal of the American Chemical Society,
1977
9.

Liotta D., Zima G.
Tetrahedron Letters,
1978
10.
10.1016/j.mencom.2022.01.026_b0050
Clive
J. Chem. Soc., Chem. Commun.,
1978
11.

Nicolaou K.C., Seitz S.P., Sipio W.J., Blount J.F.
Journal of the American Chemical Society,
1979
12.
10.1016/j.mencom.2022.01.026_b0060
Toshimitsu
J. Chem. Soc., Chem. Commun.,
1980
13.

Clive D.L., Farina V., Singh A., Wong C.K., Kiel W.A., Menchen S.M.
Journal of Organic Chemistry,
1980
14.

Toshimitsu A., Aoai T., Owada H., Uemura S., Okano M.
Journal of Organic Chemistry,
1981
15.

Alexander R.P., Paterson I.
Tetrahedron Letters,
1983
16.

Uemura S., Fukuzawa S.
Journal of the Chemical Society Perkin Transactions 1,
1985
17.

Toshimitsu A., Kusumoto T., Oida T., Tanimoto S.
Bulletin of the Chemical Society of Japan,
1991
18.

Ma S., Pan F., Hao X., Huang X.
Synlett,
2004
19.

Denmark S.E., Edwards M.G.
Journal of Organic Chemistry,
2006
20.

Alcaide B., Almendros P., Luna A., Gómez-Campillos G., Torres M.R.
Journal of Organic Chemistry,
2012
21.

Moore J.T., Soldi C., Fettinger J.C., Shaw J.T.
Chemical Science,
2013
22.

du Mont W., Kubiniok S., Peters K., von Schnering H.
Angewandte Chemie International Edition in English,
1987
23.

Du Mont W.W., Martens A., Pohl S., Saak W.
Inorganic Chemistry,
1990
24.

Salzen A.M., Meyer H.-., du Mont W.-.
Phosphorus, Sulfur and Silicon and the Related Elements,
1992
25.
10.1016/j.mencom.2022.01.026_b0125
du Mont
Main Group Chem. News,
1994
26.

du Mont W., Martens-von Salzen A., Ruthe F., Seppälä E., Mugesh G., Devillanova F.A., Lippolis V., Kuhn N.
Journal of Organometallic Chemistry,
2001
27.
10.1016/j.mencom.2022.01.026_b0135
Toshimitsu
J. Chem. Soc., Chem. Commun.,
1982
28.

Gabriele E., Singh F.V., Freudendahl D.M., Wirth T.
Tetrahedron,
2012
29.

Wang X., Li H., Zhu M., Yan J.
RSC Advances,
2017
30.
![Eigenschaften von Chalkogen–Chalkogen‐Bindungen, XI. Iod[tris(trimethylsilyl)methyl]selan: Synthese des ersten Alkylselenenyliodids](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
du Mont W., Wagner I.
Chemische Berichte,
1988
31.

Ostrowski M., Wagner I., DuMont W.-., Jones P.G., Jeske J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1993
32.

Mugesh G., Panda A., Singh H.B., Butcher R.J.
Chemistry - A European Journal,
1999
33.
10.1016/j.mencom.2022.01.026_b0165
Panda
Organometallics,
1986
34.

Mugesh G., Singh H.B., Butcher R.J.
Tetrahedron Asymmetry,
1999
35.
Saiki T., Goto K., Okazaki R.
1997
36.

Goto K., Nagahama M., Mizushima T., Shimada K., Kawashima T., Okazaki R.
Organic Letters,
2001
37.

Shimada K., Goto K., Kawashima T., Takagi N., Choe Y., Nagase S.
Journal of the American Chemical Society,
2004
38.

Shimada K., Goto K., Kawashima T.
Chemistry Letters,
2005
39.

Goto K., Sonoda D., Shimada K., Sase S., Kawashima T.
Angewandte Chemie - International Edition,
2009
40.

Sase S., Kakimoto R., Kimura R., Goto K.
Molecules,
2015
41.

Sase S., Kakimoto R., Goto K.
Angewandte Chemie - International Edition,
2014
42.

Sase S., Kimura R., Masuda R., Goto K.
New Journal of Chemistry,
2019
43.

Masuda R., Kimura R., Karasaki T., Sase S., Goto K.
Journal of the American Chemical Society,
2021
44.

Bianco A.C., Salvatore D., Gereben B., Berry M.J., Larsen P.R.
Endocrine Reviews,
2002
45.

46.

Iodide-promoted Deselenylation of β-Chloro- and β-Oxyselenides to Form Alkenes and Selenenyl Iodides
Sase S., Ebisawa K., Goto K.
Chemistry Letters,
2012
47.

Sun K., Wang X., Lv Y., Li G., Jiao H., Dai C., Li Y., Zhang C., Liu L.
Chemical Communications,
2016
48.

Sun L., Yuan Y., Yao M., Wang H., Wang D., Gao M., Chen Y., Lei A.
Organic Letters,
2019
49.

Huang B., Li Y., Yang C., Xia W.
Green Chemistry,
2020