Keywords
crystal structure
fluorescence
organoantimony compounds
rhodamine
synthesis
Abstract
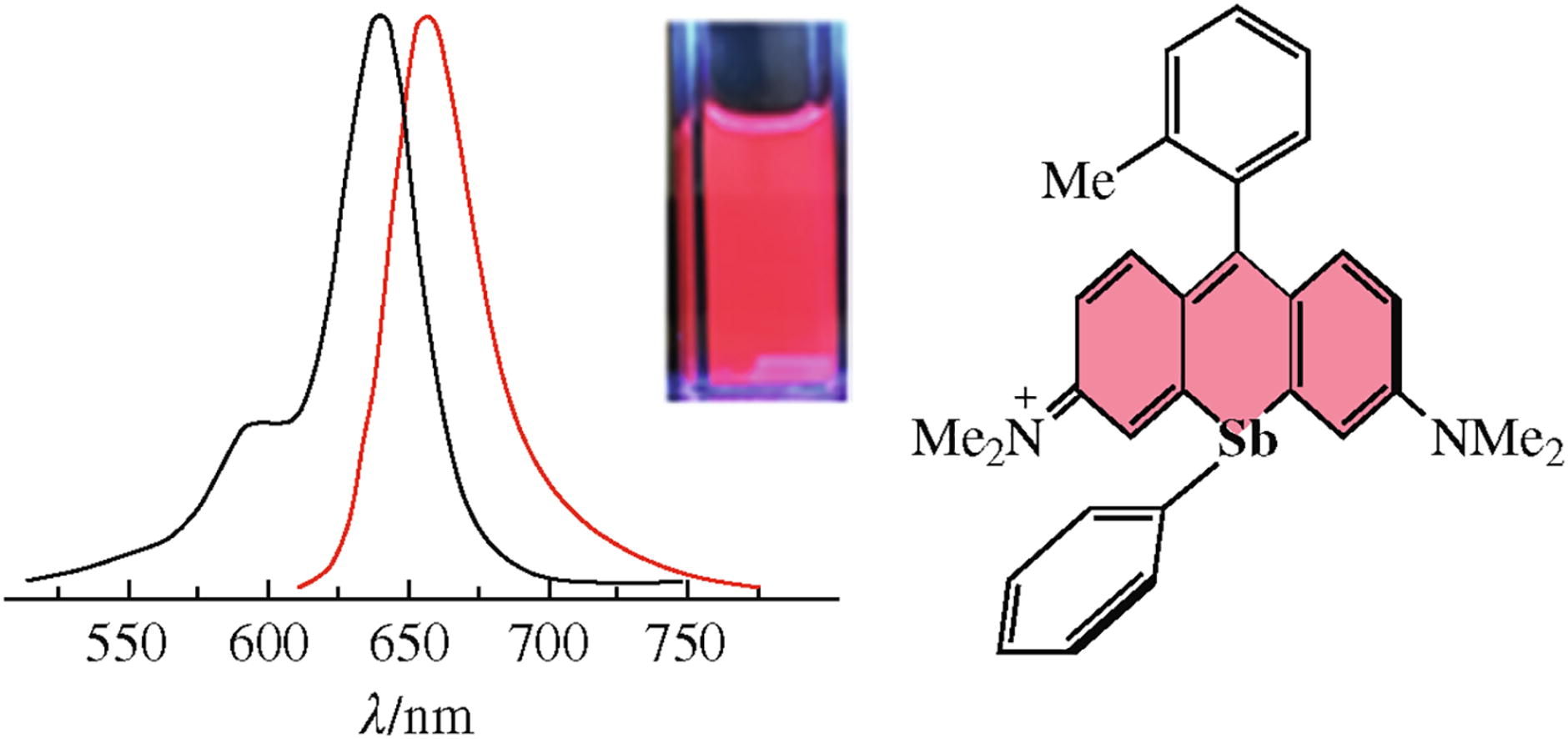
We describe the synthesis, structure and characterization of an antimony-rhodamine derivative with a phenyl-antimony moiety installed at the 10' position of the central ring. This compound is a red fluorophore (λem = 655 nm) with a quantum yield of 12% in acetonitrile.
References
1.
10.1016/j.mencom.2022.01.021_b0005
Christianson
Main Group Strategies towards Functional Hybrid Materials,
2018
2.

Hirai M., Gabbaï F.P.
Angewandte Chemie - International Edition,
2014
3.

You D., Zhou B., Hirai M., Gabbaï F.P.
Organic and Biomolecular Chemistry,
2021
4.
10.1016/j.mencom.2022.01.021_b0020
Hirai
Chem. Sci.,
1886
5.

Ke I., Myahkostupov M., Castellano F.N., Gabbaï F.P.
Journal of the American Chemical Society,
2012
6.
10.1016/j.mencom.2022.01.021_b0030
Hirai
Organometallics,
1854
7.

Christianson A.M., Gabbaï F.P.
Chemical Communications,
2017
8.

Christianson A.M., Rivard E., Gabbaï F.P.
Organometallics,
2017
9.

Christianson A.M., Gabbaï F.P.
Organometallics,
2017
10.

Ohshita J., Fujita R., Tanaka D., Ooyama Y., Kobayashi N., Higashimura H., Yamamoto Y.
Chemistry Letters,
2012
11.

Ohshita J., Yamaji K., Ooyama Y., Adachi Y., Nakamura M., Watase S.
Organometallics,
2019
12.

Li M., Li Y., Wang X., Cui X., Wang T.
Chinese Chemical Letters,
2019
13.

Deng F., Xu Z.
Chinese Chemical Letters,
2019
14.

Grzybowski M., Taki M., Senda K., Sato Y., Ariyoshi T., Okada Y., Kawakami R., Imamura T., Yamaguchi S.
Angewandte Chemie - International Edition,
2018
15.

Ikeno T., Nagano T., Hanaoka K.
Chemistry - An Asian Journal,
2017
16.

Sun Y., Liu J., Lv X., Liu Y., Zhao Y., Guo W.
Angewandte Chemie - International Edition,
2012
17.

Koide Y., Urano Y., Hanaoka K., Terai T., Nagano T.
ACS Chemical Biology,
2011
18.

Koide Y., Kawaguchi M., Urano Y., Hanaoka K., Komatsu T., Abo M., Terai T., Nagano T.
Chemical Communications,
2012
19.

Kryman M.W., Schamerhorn G.A., Hill J.E., Calitree B.D., Davies K.S., Linder M.K., Ohulchanskyy T.Y., Detty M.R.
Organometallics,
2014
20.

Hirayama T., Mukaimine A., Nishigaki K., Tsuboi H., Hirosawa S., Okuda K., Ebihara M., Nagasawa H.
Dalton Transactions,
2017
21.

Zhou X., Lai R., Beck J.R., Li H., Stains C.I.
Chemical Communications,
2016
22.
10.1016/j.mencom.2022.01.021_b0110
Sheldrick
TWINABS,
2009
23.
G. M. Sheldrick, SHELXTL, Version 6.1, Madison, WI, 1997.
24.
10.1016/j.mencom.2022.01.021_b0120
Sheldrick
SHELXL-2014, Program for Crystal Structure Refinement,
2014
25.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009