Keywords
Coordination peroxo compounds
hydrogen peroxide
lead
Organic hydroperoxides
Peroxo complexes
tin
Abstract
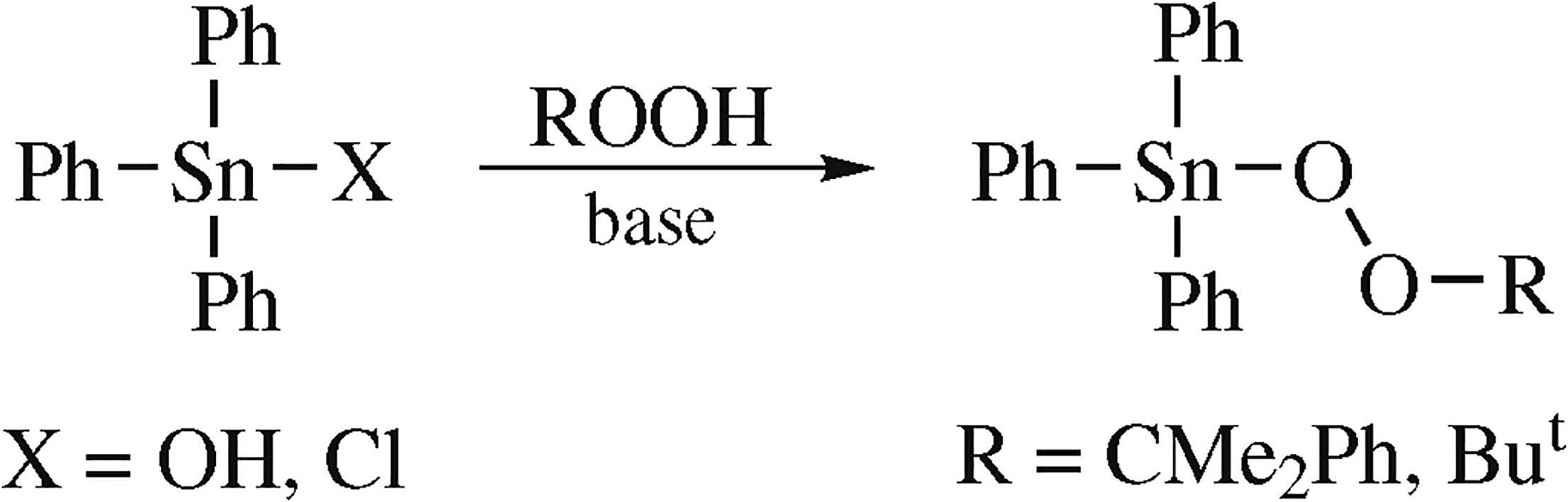
Crystalline triphenyltin cumyl and tert-butyl peroxides (Ph3SnOOCMe2Ph and Ph3SnOOBut, respectively) and triphenyllead cumyl peroxide (Ph3PbOOCMe2Ph) were synthesized and characterized by single crystal and powder X-ray diffraction, NMR, FTIR and Raman spectroscopies, TG and DSC analysis. The formation of triphenyltin tert-butyl peroxide in benzene in the presence of a base was proved by 119Sn, 13C and 1H NMR spectroscopy. To the best of our knowledge, the obtained complexes are the first structurally characterized coordination compounds of tin and lead with organic peroxides.
References
1.
10.1016/j.mencom.2022.01.018_b0005
Jones
e-EROS Encyclopedia of Reagents for Organic Synthesis,
2012
2.

Katsuki T., Sharpless K.B.
Journal of the American Chemical Society,
1980
3.
10.1016/j.mencom.2022.01.018_b0015
The Chemistry of Peroxides,
2006
4.

Heravi M.M., Zadsirjan V., Esfandyari M., Lashaki T.B.
Tetrahedron Asymmetry,
2017
5.

Sokolov N.A., Aleksandrov Y.A.
Russian Chemical Reviews,
1978
6.
10.1016/j.mencom.2022.01.018_b0030
Uhl
Ullmann’s Encyclopedia of Industrial Chemistry,
2018
7.

Bloodworth A.J., Davies A.G., Graham I.F.
Journal of Organometallic Chemistry,
1968
8.
Rieche A., Dahlmann J.
1964
9.

Salomon M.F., Salomon R.G.
Journal of the American Chemical Society,
1979
10.

Satoh W., Masumoto S., Yamamoto Y., Akiba K.
Heteroatom Chemistry,
2001
11.

Arzumanyan A.V., Novikov R.A., Terent’ev A.O., Platonov M.M., Lakhtin V.G., Arkhipov D.E., Korlyukov A.A., Chernyshev V.V., Fitch A.N., Zdvizhkov A.T., Krylov I.B., Tomilov Y.V., Nikishin G.I.
Organometallics,
2014
12.

Arzumanyan A.V., Terent'ev A.O., Novikov R.A., Lakhtin V.G., Chernyshev V.V., Fitch A.N., Nikishin G.I.
European Journal of Organic Chemistry,
2014
13.

Jana B.
Journal of Molecular Structure,
2021
14.

Uhl W., Reza Halvagar M., Claesener M.
Chemistry - A European Journal,
2009
15.

Balch A.L., Cornman C.R., Olmstead M.M.
Journal of the American Chemical Society,
1990
16.

Uhl W., Jana B.
Chemistry - A European Journal,
2008
17.

Kumar S.S., Singh S., Roesky H.W., Magull J.
Inorganic Chemistry,
2005
18.

Tsurumaki E., Sung J., Kim D., Osuka A.
Angewandte Chemie,
2016
19.
10.1016/j.mencom.2022.01.018_b0095
DiPasquale
J. Am. Chem. Soc.,
1812
20.

Mikhaylov A.A., Medvedev A.G., Churakov A.V., Grishanov D.A., Prikhodchenko P.V., Lev O.
Chemistry - A European Journal,
2016
21.

Medvedev A.G., Mikhaylov A.A., Churakov A.V., Vener M.V., Tripol’skaya T.A., Cohen S., Lev O., Prikhodchenko P.V.
Inorganic Chemistry,
2015
22.
10.1016/j.mencom.2022.01.018_b0110
Medvedev
CrystEngComm,
1922
23.

Medvedev A.G., Mikhaylov A.A., Shames A.I., Ilyukhin A.B., Churakov A.V., Grishanov D.A., Mel’nik E.A., Tripol’skaya T.A., Lev O., Prikhodchenko P.V.
Inorganic Chemistry,
2020
24.
10.1016/j.mencom.2022.01.018_b0120
Grishanov
Inorg. Chem.,
1905
25.

DiPasquale A.G., Kaminsky W., Mayer J.M.
Journal of the American Chemical Society,
2002
26.
![Non-Redox Assisted Oxygen–Oxygen Bond Homolysis in Titanocene Alkylperoxide Complexes: [Cp2TiIV(η1-OOtBu)L]+/0, L = Cl−, OTf−, Br−, OEt2, Et3P](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
DiPasquale A.G., Hrovat D.A., Mayer J.M.
Organometallics,
2006
27.

Churakov A.V., Sladkevich S., Lev O., Tripol’skaya T.A., Prikhodchenko P.V.
Inorganic Chemistry,
2010
28.

Neisen B.D., Gagnon N.L., Dhar D., Spaeth A.D., Tolman W.B.
Journal of the American Chemical Society,
2017
29.

Remarkably robust monomeric alkylperoxyzinc compounds from tris(oxazolinyl)boratozinc alkyls and O2.
Mukherjee D., Ellern A., Sadow A.D.
Journal of the American Chemical Society,
2012
30.

Coggins M.K., Martin-Diaconescu V., DeBeer S., Kovacs J.A.
Journal of the American Chemical Society,
2013
31.

Komatsuzaki H., Sakamoto N., Satoh M., Hikichi S., Akita M., Moro-oka Y.
Inorganic Chemistry,
1998
32.

Chavez F.A., Rowland J.M., Olmstead M.M., Mascharak P.K.
Journal of the American Chemical Society,
1998
33.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015