Keywords
N-heterocyclic imines
germanium
silicon
tetryliumylidenes
tin
transmetalation
Abstract
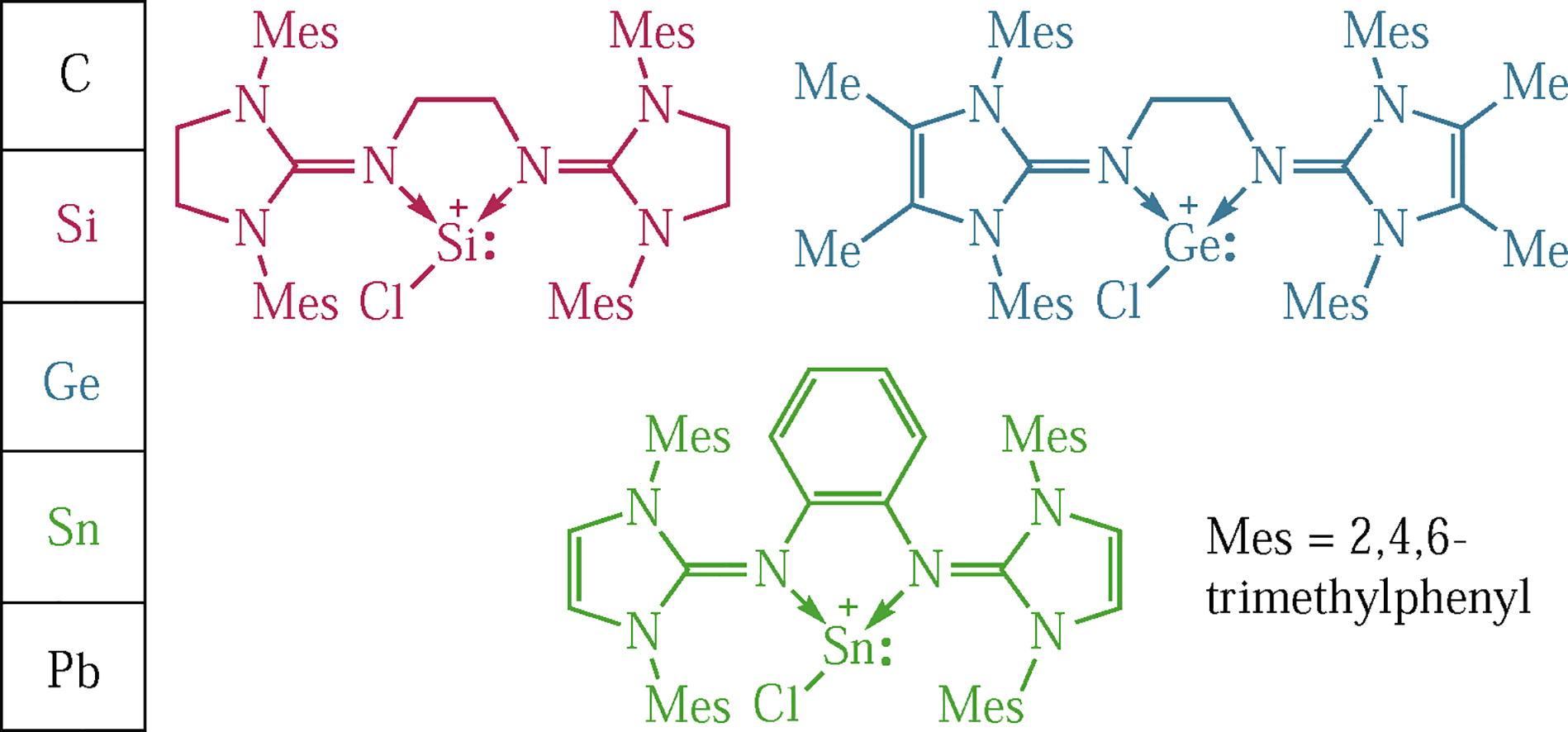
The synthesis of modified neutral bis-NHI (NHI is N-heterocyclic imine) ligands and their application for the stabilization of tetryliumylidenes are reported. The ligands’ scaffolding consists of either saturated or methylated imidazoline backbones, and the bridge alternated from flexible ethylene to more rigid o-phenylene. Transmetalation reactivity of the cationic SnII compounds was tested towards LiAlH4 and IDipp→SiCl2 [IDipp is 1,3-bis(2,6-diisopropyl- phenyl)imidazol-2-ylidene] affording the respective aluminium and silicon complexes.
References
1.

Wang Y., Karni M., Yao S., Apeloig Y., Driess M.
Journal of the American Chemical Society,
2018
2.

Zhou Y., Karni M., Yao S., Apeloig Y., Driess M.
Angewandte Chemie - International Edition,
2016
3.

Yao S., Xiong Y., Driess M.
Accounts of Chemical Research,
2017
4.

Tschernuth F.S., Hanusch F., Szilvási T., Inoue S.
Organometallics,
2020
5.

Ochiai T., Franz D., Inoue S.
Chemical Society Reviews,
2016
6.

Doddi A., Peters M., Tamm M.
Chemical Reviews,
2019
7.

Hanusch F., Munz D., Sutter J., Meyer K., Inoue S.
Angewandte Chemie - International Edition,
2021
8.

Majhi P.K., Sasamori T.
Chemistry - A European Journal,
2018
9.

Frisch P., Inoue S.
Dalton Transactions,
2020
10.

Frisch P., Szilvási T., Porzelt A., Inoue S.
Inorganic Chemistry,
2019
11.

Yeong H., Li Y., So C.
Organometallics,
2014
12.

Swamy V.S., Pal S., Khan S., Sen S.S.
Dalton Transactions,
2015
13.

Fritz-Langhals E.
Organic Process Research and Development,
2019
14.
Powley S.L., Hanusch F., Inoue S.
2020
15.

Nesterov V., Reiter D., Bag P., Frisch P., Holzner R., Porzelt A., Inoue S.
Chemical Reviews,
2018
16.

Börner J., Flörke U., Glöge T., Bannenberg T., Tamm M., Jones M.D., Döring A., Kuckling D., Herres-Pawlis S.
Journal of Molecular Catalysis A Chemical,
2010
17.

Glöge T., Petrovic D., Hrib C., Daniliuc C., Herdtweck E., Jones P., Tamm M.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2010
18.

Neuba A., Herres-Pawlis S., Flörke U., Henkel G.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2008
19.
![[Bis(guanidine)]zinc Complexes and Their Application in Lactide Polymerisation](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Börner J., Herres‐Pawlis S., Flörke U., Huber K.
European Journal of Inorganic Chemistry,
2007
20.

Denker L., Trzaskowski B., Frank R.
Chemical Communications,
2021
21.

Franz D., Irran E., Inoue S.
Angewandte Chemie - International Edition,
2014
22.

Franz D., Szilvási T., Pöthig A., Deiser F., Inoue S.
Chemistry - A European Journal,
2018
23.

Volbeda J., Jones P.G., Tamm M.
Inorganica Chimica Acta,
2014
24.

Singh A.P., Roesky H.W., Carl E., Stalke D., Demers J., Lange A.
Journal of the American Chemical Society,
2012
25.

Muhr M., Heiß P., Schütz M., Bühler R., Gemel C., Linden M.H., Linden H.B., Fischer R.A.
Dalton Transactions,
2021
26.
APEX Suite of Crystallographic Software, Bruker AXS, Madison, WI, 2015.
27.
SAINT, version 7.56a, and SADABS, version 2008/1, Bruker AXS, Madison, WI, 2008.
28.
10.1016/j.mencom.2022.01.004_b0065
Sheldrick
SHELXL-2014,
2014
29.

Hübschle C.B., Sheldrick G.M., Dittrich B.
Journal of Applied Crystallography,
2011
30.
10.1016/j.mencom.2022.01.004_b0075
Sheldrick
SHELXL-97,
1998