Keywords
anilines
copper catalysts
hydrogen
hydrogenation
nitro group
nitroarenes
Abstract
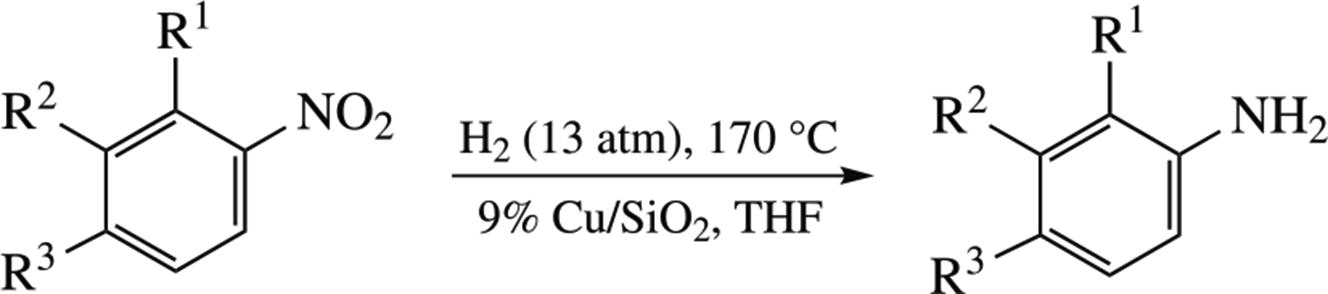
A 9% Cu/SiO2 catalyst prepared by the deposition– precipitation technique was active in the hydrogenation of substituted nitrobenzenes at 170 °C and 13 atm H2 in a batch reactor. Nitro groups were selectively reduced to NH2 groups except for the case of 5-bromo-1,3-dinitrobenzene. The selectivity of the overall process is somewhat suppressed by inevitable formation of azo compounds and traces of tars due to the reactions of the obtained amines with nitroso intermediates.
References
1.

Song J., Huang Z., Pan L., Li K., Zhang X., Wang L., Zou J.
Applied Catalysis B: Environmental,
2018
2.

Kadam H.K., Tilve S.G.
RSC Advances,
2015
3.

Formenti D., Ferretti F., Scharnagl F.K., Beller M.
Chemical Reviews,
2018
4.

Couto C.S., Madeira L.M., Nunes C.P., Araújo P.
Chemical Engineering and Technology,
2015
5.

Jaf Z.N., Altarawneh M., Miran H.A., Almatarneh M.H., Jiang Z., Dlugogorski B.Z.
ACS Omega,
2018
6.

Tomkins P., Gebauer-Henke E., Leitner W., Müller T.E.
ACS Catalysis,
2014
7.

Monti G.A., Correa N.M., Falcone R.D., Silbestri G.F., Moyano F.
RSC Advances,
2020
8.
Nuzhdin A.L., Bukhtiyarova G.A., Lin T., Gerasimov E.Y., Bukhtiyarov V.I.
Mendeleev Communications,
2019
9.

Campos C., Torres C., Oportus M., Peña M.A., Fierro J.L., Reyes P.
Catalysis Today,
2013
10.
10.1016/j.mencom.2021.11.036_b0050
Litvin
Ross. Khim. Zh.,
2000
11.

Shuvalova E.V., Kirichenko O.A., Kustov L.M.
Russian Chemical Bulletin,
2017
12.

Varkolu M., Velpula V., Pochamoni R., Muppala A.R., Burri D.R., Kamaraju S.R.
Applied Petrochemical Research,
2015
13.

Huang L., Lv Y., Wu S., Liu P., Xiong W., Hao F., Luo H.
Applied Catalysis A: General,
2019
14.

Tejeda-Serrano M., Cabrero-Antonino J.R., Mainar-Ruiz V., López-Haro M., Hernández-Garrido J.C., Calvino J.J., Leyva-Pérez A., Corma A.
ACS Catalysis,
2017
15.

Cheong W., Yang W., Zhang J., Li Y., Zhao D., Liu S., Wu K., Liu Q., Zhang C., Wang D., Peng Q., Chen C., Li Y.
ACS applied materials & interfaces,
2019
16.

Shesterkina A.A., Strekalova A.A., Kustov L.M.
Russian Journal of Physical Chemistry A,
2020
17.

Kour G., Gupta M., Vishwanathan B., Thirunavukkarasu K.
New Journal of Chemistry,
2016
18.

Ye T., Lu Y., Li J., Nakao T., Yang H., Tada T., Kitano M., Hosono H.
Journal of the American Chemical Society,
2017
19.

Shuvalova E.V., Kirichenko O.A., Kapustin G.I., Kustov L.M.
Russian Chemical Bulletin,
2016
20.

Kirichenko O.A., Shuvalova E.V., Strekalova A.A., Davshan N.A., Kapustin G.I., Nissenbaum V.D.
Russian Journal of Physical Chemistry A,
2018
21.

Kirichenko O.A., Shuvalova E.V., Redina E.A.
Russian Chemical Bulletin,
2019
22.

Xu C., Chen G., Zhao Y., Liu P., Duan X., Gu L., Fu G., Yuan Y., Zheng N.
Nature Communications,
2018
23.

Van Der Grift C.J., Wielers A.F., Mulder A., Geus J.W.
Thermochimica Acta,
1990
24.

Zhang H., Tan H., Jaenicke S., Chuah G.
Journal of Catalysis,
2020