Keywords
biologically active compounds
carbon nanoparticles
controlled release
delivery systems
dextran sulfate sodium salt
encapsulation
enzyme activity
infrared laser
microchamber arrays
Abstract
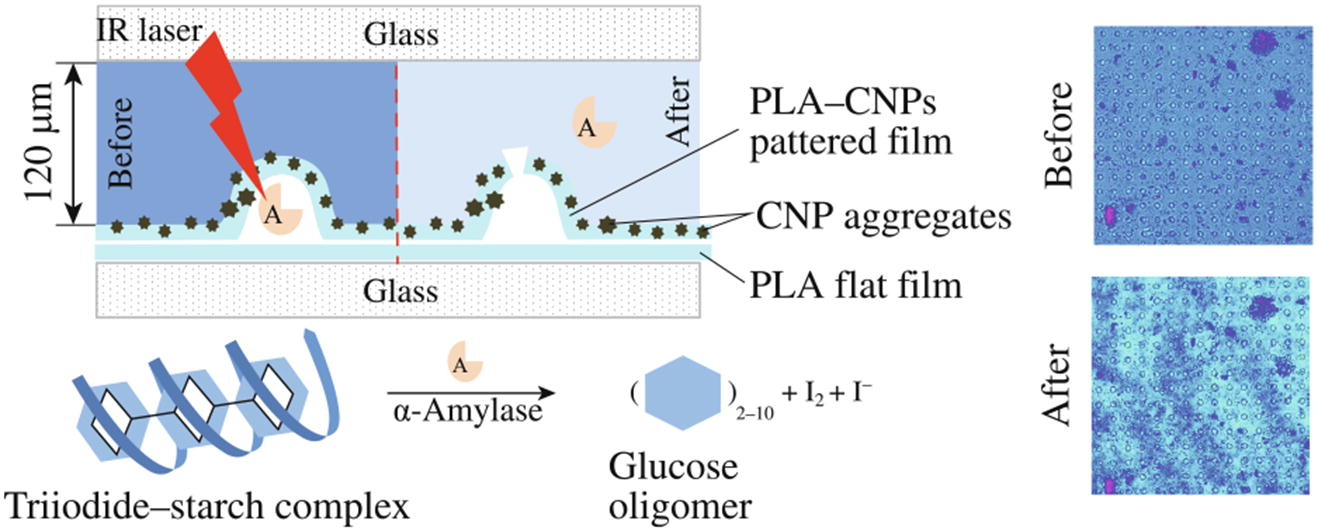
Microchamber arrays have been prepared from a composite of polylactic acid and carbon nanoparticles. Due to the presence of the nanoparticles, the microchambers are sensitive to infrared laser irradiation. α-Amylase has been encapsulated in the microchambers and released under controlled laser irradiation at 1064 nm with retention of the enzyme activity.
References
1.
Tiwari G., Tiwari R., Bannerjee S., Bhati L., Pandey S., Pandey P., Sriwastawa B.
International Journal of Pharmaceutical Investigation,
2012
2.

De Koker S., Hoogenboom R., De Geest B.G.
Chemical Society Reviews,
2012
3.

Timko B.P., Dvir T., Kohane D.S.
Advanced Materials,
2010
4.

Fu X., Hosta-Rigau L., Chandrawati R., Cui J.
Chem,
2018
5.

Gao H., Goriacheva O.A., Tarakina N.V., Sukhorukov G.B.
ACS applied materials & interfaces,
2016
6.

Bogdanova L.R., Makarova A.O., Zueva O.S., Zakharova L.Y., Zuev Y.F.
Russian Chemical Bulletin,
2020
7.
Anakhov M.V., Gumerov R.A., Potemkin I.I.
Mendeleev Communications,
2020
8.

Sindeeva O.A., Prikhozhdenko E.S., Bratashov D.N., Vostrikova A.M., Atkin V.S., Ermakov A.V., Khlebtsov B.N., Sapelkin A.V., Goryacheva I.Y., Sukhorukov G.B.
Soft Matter,
2018
9.

Kiryukhin M.V., Lau H.H., Goh S.H., Teh C., Korzh V., Sadovoy A.
Talanta,
2018
10.

Kopach O., Zheng K., Sindeeva O.A., Gai M., Sukhorukov G.B., Rusakov D.A.
Biomaterials Science,
2019
11.

Sindeeva O.A., Gusliakova O.I., Inozemtseva O.A., Abdurashitov A.S., Brodovskaya E.P., Gai M., Tuchin V.V., Gorin D.A., Sukhorukov G.B.
ACS applied materials & interfaces,
2018
12.
10.1016/j.mencom.2021.11.034_b0060
Sindeeva
Biotechnol.,
2020
13.

Zykova Y., Kudryavtseva V., Gai M., Kozelskaya A., Frueh J., Sukhorukov G., Tverdokhlebov S.
European Polymer Journal,
2019
14.

Kane B.J., Younan G., Helm D., Dastouri P., Prentice-Mott H., Irimia D., Chan R.K., Toner M., Orgill D.P.
Journal of Biomedical Materials Research - Part A,
2010
15.

Yoo D., Lee J., Shin T., Cheon J.
Accounts of Chemical Research,
2011
16.
10.1016/j.mencom.2021.11.034_b0080
Hayashi
Mater. Interfaces,
1903
17.

Huang S., Liu J., He Q., Chen H., Cui J., Xu S., Zhao Y., Chen C., Wang L.
Nano Research,
2015
18.
10.1016/j.mencom.2021.11.034_b0090
Huang
Nano Res.,
1932
19.

Microchamber arrays made of biodegradable polymers for enzymatic release of small hydrophilic cargos
Zhang J., Sun R., DeSouza-Edwards A.O., Frueh J., Sukhorukov G.B.
Soft Matter,
2020
20.
10.1016/j.mencom.2021.11.034_b0100
Ash
Lasers Med. Sci.,
1909
21.

Zhao Z., Fairchild P.W.
Proceedings of SPIE - The International Society for Optical Engineering,
1998
22.

Clement M., Daniel G., Trelles M.
Journal of Cosmetic and Laser Therapy,
2005
23.

GHOSH P., HAN G., DE M., KIM C., ROTELLO V.
Advanced Drug Delivery Reviews,
2008
24.

Zhou F., Wu S., Wu B., Chen W.R., Xing D.
Small,
2011
25.

Kam N.W., O'Connell M., Wisdom J.A., Dai H.
Proceedings of the National Academy of Sciences of the United States of America,
2005
26.

Li W., Wang J., Ren J., Qu X.
Angewandte Chemie,
2013
27.

Li J., Tang B., Yuan B., Sun L., Wang X.
Biomaterials,
2013
28.

Dekaliuk M.O., Viagin O., Malyukin Y.V., Demchenko A.P.
Physical Chemistry Chemical Physics,
2014
29.

Baker S., Baker G.
Angewandte Chemie - International Edition,
2010
30.

Li H., Kang Z., Liu Y., Lee S.
Journal of Materials Chemistry A,
2012
31.

Kokorina A.A., Prikhozhdenko E.S., Tarakina N.V., Sapelkin A.V., Sukhorukov G.B., Goryacheva I.Y.
Carbon,
2018
32.

Huggins C., Russell P.S.
Annals of Surgery,
2008
33.

Pimstone N.R.
Clinical Chemistry,
1964
34.

Cochran B., Lunday D., Miskevich F.
Journal of Chemical Education,
2008