Keywords
antitumor activity
benzofuroxans
hybrid compounds
imidazol-2-ones
regioselective synthesis
SNAr reaction
Abstract
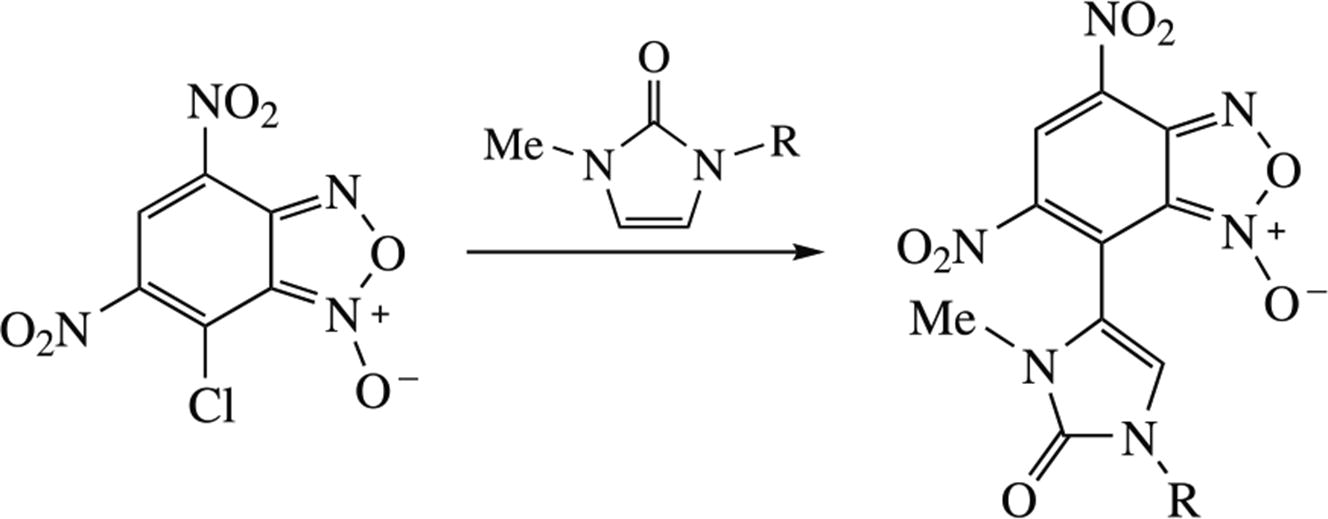
Novel hybrid compounds containing both benzofuroxan and 1H-imidazol-2(3H)-one moieties were synthesized by an SNAr reaction between 7-chloro-4,6-dinitrobenzofuroxan and imidazol-2-ones, with the only C4 regioisomer having been formed. Cytotoxic effects of parent and obtained compounds were estimated on human cancer and normal cells, while two of imidazolones showed higher cytotoxicity in relation to the M-HeLa cancer line compared to the hybrid products. In relation to the normal liver cell line Chang, the tested compounds were found to be non-toxic and can be promising for further development of anticancer agents.
References
1.

Han S., Siegel D.S., Morrison K.C., Hergenrother P.J., Movassaghi M.
Journal of Organic Chemistry,
2013
2.

Shaw A.W., Paone D.V., Nguyen D.N., Stump C.A., Burgey C.S., Mosser S.D., Salvatore C.A., Rutledge R.Z., Kane S.A., Koblan K.S., Graham S.L., Vacca J.P., Williams T.M.
Bioorganic and Medicinal Chemistry Letters,
2007
3.

Watanabe K., Morinaka Y., Hayashi Y., Shinoda M., Nishi H., Fukushima N., Watanabe T., Ishibashi A., Yuki S., Tanaka M.
Bioorganic and Medicinal Chemistry Letters,
2008
4.
10.1016/j.mencom.2021.11.032_b0020
Comprehensive Natural Products III,
2020
5.
10.1016/j.mencom.2021.11.032_b0025
Bunag
in xPharm: The Comprehensive Pharmacology Reference, eds,
2007
6.
10.1016/j.mencom.2021.11.032_b0030
Davis
in xPharm: The Comprehensive Pharmacology Reference, eds,
2007
7.

Sheppeck J.E., Gilmore J.L., Tebben A., Xue C., Liu R., Decicco C.P., Duan J.J.
Bioorganic and Medicinal Chemistry Letters,
2007
8.
10.1016/j.mencom.2021.11.032_b0040
Kitazaki
Chem. Pharm. Bull.,
1935
9.

Bronson J.J., DenBleyker K.L., Falk P.J., Mate R.A., Ho H., Pucci M.J., Snyder L.B.
Bioorganic and Medicinal Chemistry Letters,
2003
10.

Xue N., Yang X., Wu R., Chen J., He Q., Yang B., Lu X., Hu Y.
Bioorganic and Medicinal Chemistry,
2008
11.

Congiu C., Cocco M.T., Onnis V.
Bioorganic and Medicinal Chemistry Letters,
2008
12.

Zav'yalov S.I., Ezhova G.I., Kravchenko N.E., Kulikova L.B., Dorofeeva O.V., Rumyantseva E.E., Zavozin A.G.
Pharmaceutical Chemistry Journal,
2004
13.

Antonova M.M., Baranov V.V., Kravchenko A.N.
Chemistry of Heterocyclic Compounds,
2015
14.

Das S., Fröhlich R., Pramanik A.
Organic Letters,
2006
15.

Diness F., Meldal M.
Biopolymers,
2010
16.

Chugunova E., Samsonov V., Gerasimova T., Rybalova T., Bagryanskaya I.
Tetrahedron,
2015
17.

Smolobochkin A., Gazizov A., Sazykina M., Akylbekov N., Chugunova E., Sazykin I., Gildebrant A., Voronina J., Burilov A., Karchava S., Klimova M., Voloshina A., Sapunova A., Klimanova E., Sashenkova T., et. al.
Molecules,
2019
18.

Claudio Viegas-Junior, Eliezer J. Barreiro, Carlos Alberto Manssour Fraga
Current Medicinal Chemistry,
2007
19.

Chugunova E.A., Akylbekov N.I., Appazov N.O., Makhrus E.M., Burilov A.R.
Russian Journal of Organic Chemistry,
2016
20.

Serkov I.V., Chugunova E.A., Burilov A.R., Bachurin S.O.
Doklady Chemistry,
2013
21.
Burilov A.R., Khakimov M.S., Gazizov A.S., Pudovik M.A., Syakaev V.V., Krivolapov D.B., Konovalov A.I.
Mendeleev Communications,
2008
22.

Rodriguez-Dafonte P., Terrier F., Lakhdar S., Kurbatov S., Goumont R.
Journal of Organic Chemistry,
2009
23.

Kurbatov S., Rodriguez-Dafonte P., Goumont R., Terrier F.
Chemical Communications,
2003
24.

Micheletti G., Bordoni S., Chugunova E., Boga C.
Molecules,
2017
25.

Llopart C.C., Ferrer C., Joule J.A.
Canadian Journal of Chemistry,
2004
26.

Chugunova E., Akylbekov N., Shakirova L., Dobrynin A., Syakaev V., Latypov S., Bukharov S., Burilov A.
Tetrahedron,
2016
27.

Voloshina A.D., Sapunova A.S., Kulik N.V., Belenok M.G., Strobykina I.Y., Lyubina A.P., Gumerova S.K., Kataev V.E.
Bioorganic and Medicinal Chemistry,
2021