Keywords
[5+1] heterocyclization
in vitro antibacterial screening
cross-imidination
formamidines
microwave-assisted reactions
multicomponent reactions
pyrimidinones
tandem reactions
thieno[2,3-b]pyridines
Abstract
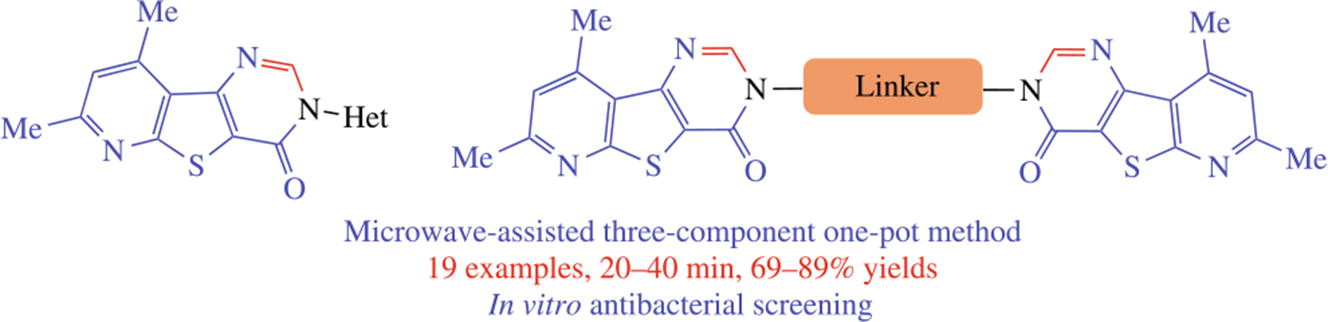
A three-component protocol involving the reaction of 3-aminothieno[2,3-b]pyridine-2-carboxylate, Me2NCH(OMe)2 and heteroaryl (bis)amines in dioxane under microwave irradiation yielded a new series of pyrimidinones. The target hybrids were formed by an initial formamidine formation,Pyrazole-linked pyrimidinones displayed the best antibacterial activity against all the gram-positive and negative strains tested.
References
1.
![Synthesis, Characterization, and Antimicrobial Evaluation of Novel Thiohydrazonates and Pyrazolo[3,4-b]pyridines](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mekky A.E., Sanad S.M.
Polycyclic Aromatic Compounds,
2019
2.

Sanad S.M., Mekky A.E., Said A.Y., Elneairy M.A.
Journal of Heterocyclic Chemistry,
2021
3.
10.1016/j.mencom.2021.11.031_b0015
Sanad
Synth. Commun.,
1982
4.

Amin L.H., Shawer T.Z., El-Naggar A.M., El-Sehrawi H.M.
Bioorganic Chemistry,
2019
5.

Kostova I., Atanasov P.Y.
Current Organic Chemistry,
2017
6.

Farghaly A.M., AboulWafa O.M., Elshaier Y.A., Badawi W.A., Haridy H.H., Mubarak H.A.
Medicinal Chemistry Research,
2019
7.

Bai S., Liu S., Zhu Y., Wu Q.
Tetrahedron Letters,
2018
8.

Hese S.V., Meshram R.J., Kamble R.D., Mogle P.P., Patil K.K., Kamble S.S., Gacche R.N., Dawane B.S.
Medicinal Chemistry Research,
2017
9.

Rashid H.U., Martines M.A., Duarte A.P., Jorge J., Rasool S., Muhammad R., Ahmad N., Umar M.N.
RSC Advances,
2021
10.

Sanad S.M., Ahmed A.A., Mekky A.E.
Journal of Heterocyclic Chemistry,
2019
11.

Maurya S.S., Bahuguna A., Khan S.I., Kumar D., Kholiya R., Rawat D.S.
European Journal of Medicinal Chemistry,
2019
12.
Abdelhamid I.A., Hawass M.A., Sanad S.M., Elwahy A.H.
Arkivoc,
2021
13.
![Efficient Synthesis and Characterization of Novel Pyrido[3′,2′:4,5]thieno[3,2‐ d ]pyrimidines and Their Fused [1,2,4]triazole Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Hawass M.A., Ahmed A.A., Elneairy M.A.
Journal of Heterocyclic Chemistry,
2018
14.

Zheng G.Z., Bhatia P., Daanen J., Kolasa T., Patel M., Latshaw S., El Kouhen O.F., Chang R., Uchic M.E., Miller L., Nakane M., Lehto S.G., Honore M.P., Moreland R.B., Brioni J.D., et. al.
Journal of Medicinal Chemistry,
2005
15.
![Synthesis and Biological Activity of Pyrido[3′,2′:4,5]thieno[3,2-d]pyrimidines as Phosphodiesterase Type 4 Inhibitors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Taltavull J., Serrat J., Gràcia J., Gavaldà A., Andrés M., Córdoba M., Miralpeix M., Vilella D., Beleta J., Ryder H., Pagès L.
Journal of Medicinal Chemistry,
2010
16.
![Synthesis and antimicrobial activity of new derivatives of pyrano[4'',3'':4',5']pyrido[3',2':4,5]thieno[3,2-d]pyrimidine and new heterocyclic systems](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Sirakanyan S.N., Spinelli D., Geronikaki A., Kartsev V.G., Hakobyan E.K., Hovakimyan A.A.
Synthetic Communications,
2019
17.
![Microwave-Assisted Synthesis of Potential Bioactive Benzo-, Pyrido- or Pyrazino-thieno[3,2-d]pyrimidin-4-amine Analogs of MPC-6827](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Loidreau Y., Nourrisson M., Fruit C., Corbière C., Marchand P., Besson T.
Pharmaceuticals,
2020
18.
![Synthesis, Antitumor Activity, and Docking Analysis of New Pyrido[3’,2’:4,5]furo(thieno)[3,2-d]pyrimidin-8-amines](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Sirakanyan, Spinelli, Geronikaki, Hakobyan, Sahakyan, Arabyan, Zakaryan, Nersesyan, Aharonyan, Danielyan, Muradyan, Hovakimyan
Molecules,
2019
19.
![Regioselective synthesis and theoretical calculations of Bis(pyrido[2′,3′:3,4]pyrazolo[1,5-a]pyrimidines) linked to benzofuran units via piperazine spacer: A DFT, MM2, and MMFF94 study](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sanad S.M., Ahmed M.S., Mekky A.E., Abdallah Z.A.
Journal of Molecular Structure,
2021
20.
10.1016/j.mencom.2021.11.031_b0100
Hawass
Synth. Commun.,
1899
21.

Dyachenko I.V., Dyachenko V.D., Dorovatovskii P.V., Khrustalev V.N., Nenajdenko V.G.
Chemistry of Heterocyclic Compounds,
2020
22.
![On the reactivity of pyrido[3′,2′:4,5]furo(thieno)[3,2-d]pyrimidin-7(8)-ones with some alkyl mono- and di-halides: synthesis of new heterocyclic systems containing thiazolo[3,2-a]pyrimidine and pyrimido[2,1-b]thiazine moiety](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sirakanyan S.N., Spinelli D., Geronikaki A., Hovakimyan A.A.
Tetrahedron,
2015
23.
10.1016/j.mencom.2021.11.031_b0115
Bohm
Pharmazie,
1992
24.

Elnagdi M.H., Erian A.W.
Archiv der Pharmazie,
1991
25.

Shaaban M.R., Saleh T.S., Mayhoub A.S., Mansour A., Farag A.M.
Bioorganic and Medicinal Chemistry,
2008
26.
Sanad S.M., Mekky A.E., Said A.Y., Elneairy M.A.
Mendeleev Communications,
2021
27.

Mohammad H., Reddy P.V., Monteleone D., Mayhoub A.S., Cushman M., Seleem M.N.
European Journal of Medicinal Chemistry,
2015