Keywords
aldol condensation
enynals
heterocyclization
imidazolidines
organogermanium compounds
oxazoles
Α-enals
Abstract
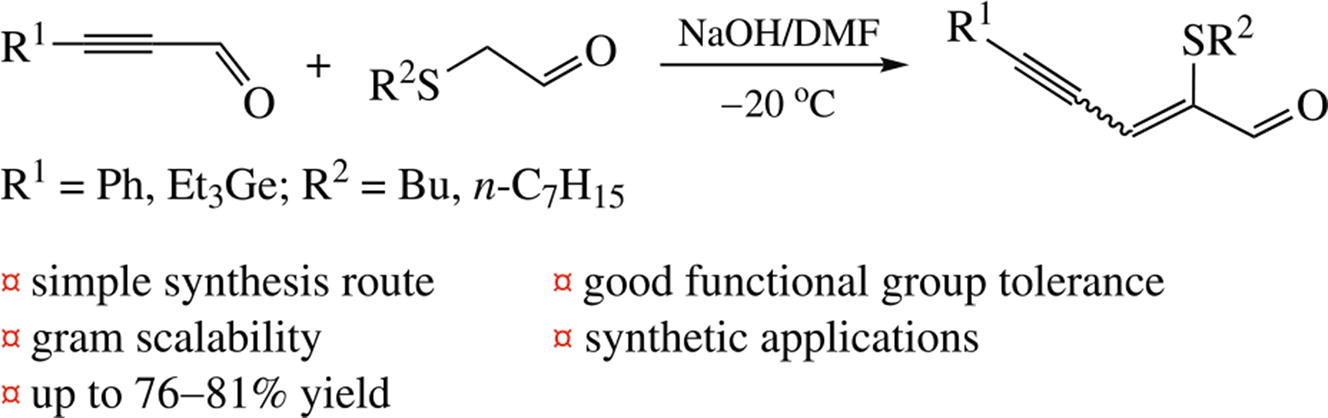
An efficient synthesis of new (Z,E)-2-(alkylthio)alk-2-en-4-ynals by aldol condensation reaction of ynals with (alkylthio)-acetaldehydes using NaOH/DMF system has been developed.The reaction of these products with N,N-diphenylethylene-diamine and p-tosylmethyl isocyanide proceeds at the carbonyl group to form the corresponding imidazolidine and oxazole derivatives.
References
1.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
2.

Keiko N.A., Vchislo N.V.
Asian Journal of Organic Chemistry,
2016
3.

Vchislo N.V.
Asian Journal of Organic Chemistry,
2019
4.

Vinogradov M.G., Turova O.V., Zlotin S.G.
Organic and Biomolecular Chemistry,
2019
5.

Reyes-Rodríguez G.J., Rezayee N.M., Vidal-Albalat A., Jørgensen K.A.
Chemical Reviews,
2019
6.

Vchislo N.V., Verochkina E.A.
ChemistrySelect,
2020
7.

Golovanov A.A., Odin I.S., Zlotskii S.S.
Russian Chemical Reviews,
2019
8.

Sokov S.A., Odin I.S., Gusev D.M., Kunavin Y.A., Vologzhanina A.V., Voronova E.D., Golovanov A.A.
Russian Chemical Bulletin,
2020
9.
(c) A. A. Golovanov, D. M. Gusev, I. S. Odin and S. S. Zlotskii, Chem. Heterocycl. Compd., 2019, 55, 333 (Khim. Geterotsikl. Soedin., 2019, 55, 333)
10.

Igushkina A.V., Golovanov A.A., Boyarskaya I.A., Kolesnikov I.E., Vasilyev A.V.
Molecules,
2020
11.

Huang J., Li F., Cui L., Su S., Jia X., Li J.
Chemical Communications,
2020
12.

Vila C., Cernicharo‐Toledo F., Blay G., Pedro J.R.
European Journal of Organic Chemistry,
2021
13.

Hoffman R.V., Shechter H.
Journal of the American Chemical Society,
1978
14.

Hoffman R.V., Orphanides G.G., Shechter H.
Journal of the American Chemical Society,
1978
15.

Hoffman H.M., Vinter J.G.
Journal of Organic Chemistry,
1974
16.

Hoffman R.V., Shechter H.
Journal of the American Chemical Society,
1971
17.

Buser D., Pauling H., Thum A., Bonrath W.
Molecules,
2002
18.

Sun J., Zheng G., Fu Y., Wang L., Li Y., Zhang Q.
Organic Letters,
2018
19.

Mandal S., Mandal S., Ghosh S.K., Ghosh A., Saha R., Banerjee S., Saha B.
Synthetic Communications,
2016
20.
A. A. Golovanov, D. R. Latypov, V. V. Bekina, V. S. Pisareva, A. V. Vologzhanina and V. A. Dokichev, Russ. J. Org. Chem., 2013, 49, 1264 (Zh. Org. Khim., 2013, 49, 1282).
21.
Keiko N.A., Stepanova L.G., Verochkina E.A., Larina L.I.
Arkivoc,
2010
22.
(a) A. S. Medvedeva, Russ. J. Org. Chem., 1996, 32, 272 (Zh. Org.Khim., 1996, 32, 289)
23.
Elshina V.G., Novokshonov V.V., Verochkina E.A., Ushakov I.A., Rosentsveig I.B., Vchislo N.V.
Mendeleev Communications,
2019
24.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Jr. Montgomery, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Lyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision C.01, Gaussian, Wallingford, CT, 2010.
25.

Domingo L.R., Pérez P., Sáez J.A.
RSC Advances,
2013
26.

Zhang H., Zhao Z., Zhou C.
European Journal of Medicinal Chemistry,
2018
27.

Mhlongo J.T., Brasil E., de la Torre B.G., Albericio F.
Marine Drugs,
2020
28.

Ke S., Zhang Z., Shi L., Liu M., Fang W., Zhang Y., Wu Z., Wan Z., Long T., Wang K.
Organic and Biomolecular Chemistry,
2019
29.

Swain S.P., Mohanty S.
ChemMedChem,
2019
30.

Husain A., Ahmad A., Khan S.A., Asif M., Bhutani R., Al-Abbasi F.A.
Saudi Pharmaceutical Journal,
2016
31.

Moussa I.A., Banister S.D., Manoli M., Doddareddy M.R., Cui J., Mach R.H., Kassiou M.
Bioorganic and Medicinal Chemistry Letters,
2012
32.

Tarannum S., Sk S., Das S., Wani I.A., Ghorai M.K.
Journal of Organic Chemistry,
2019
33.

Golovanov A.A., Bekin V.V., Odin I.S., Chertov A.Y., Grigor’eva O.B., Pisareva V.S.
Russian Journal of Organic Chemistry,
2015