Keywords
methyl aluminum complexes
organoaluminum compounds
ring-opening of thiiranes
ring-opening polymerization of lactones
SNS-polydentate ligands
thiiranes
Abstract
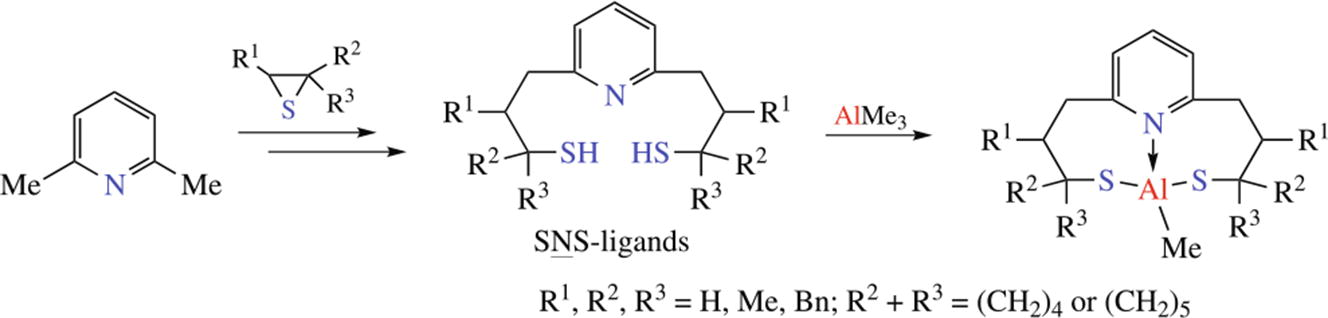
New SNS-ligands were obtained by consequent ring-opening of substituted thiiranes by lithiated 2,6-lutidine, mono- functionalized SN-ligands having been isolated as the intermediate compounds. The molecular structure of ligand 2,6-Py(CH2CH2CBn2SH)2 was elucidated by XRD analysis.The reaction of AlMe3 with SNS-ligands afforded monomeric methyl aluminum complexes which have been tested in ring- opening polymerization of ε caprolactone in bulk.
References
1.
10.1016/j.mencom.2021.11.026_b0005
Lawrance
Introduction to Coordination Chemistry,
2013
2.

Kuchuk E.A., Mankaev B.N., Zaitsev K.V., Oprunenko Y.F., Churakov A.V., Zaitseva G.S., Karlov S.S.
Inorganic Chemistry Communication,
2016
3.

Kellogg R.M.
Phosphorus, Sulfur and Silicon and the Related Elements,
1997
4.

Pellissier H.
Tetrahedron,
2007
5.

Zaitsev K.V., Bermeshev M.V., Samsonov A.A., Oprunenko J.F., Churakov A.V., Howard J.A., Karlov S.S., Zaitseva G.S.
New Journal of Chemistry,
2008
6.

Kireenko M.M., Kuchuk E.A., Zaitsev K.V., Tafeenko V.A., Oprunenko Y.F., Churakov A.V., Lermontova E.K., Zaitseva G.S., Karlov S.S.
Dalton Transactions,
2015
7.

Zaitsev K.V., Cherepakhin V.S., Zherebker A., Kononikhin A., Nikolaev E., Churakov A.V.
Journal of Organometallic Chemistry,
2018
8.

Zaitsev K.V., Cherepakhin V.S., Churakov A.V., Peregudov A.S., Tarasevich B.N., Egorov M.P., Zaitseva G.S., Karlov S.S.
Inorganica Chimica Acta,
2016
9.

Cherepakhin V.S., Zaitsev K.V., Oprunenko Y.F., Churakov A.V., Lermontova E.K., Zaitseva G.S., Karlov S.S.
European Journal of Inorganic Chemistry,
2015
10.
10.1016/j.mencom.2021.11.026_b0050
Kaptein
J. Org. Chem.,
1890
11.
Koning B., Boer J.H., Meetsma A., Kellogg R.M.
Arkivoc,
2004
12.

Ongoka P., Mauzé B., Miginiac L.
Synthesis,
1985
13.

Braanalt J., Kvarnstroem I., Svensson S.C., Classon B., Samuelsson B.
Journal of Organic Chemistry,
1994
14.

Mountford S.J., Campi E.M., Robinson A.J., Hearn M.T.
Tetrahedron,
2011
15.

Bordwell F.G., Andersen H.M., Pitt B.M.
Journal of the American Chemical Society,
1954
16.

17.

18.

Adam W., Bargon R.M.
Chemical Reviews,
2003
19.

Yadav L.D., Kapoor R.
Synthesis,
2002
20.

Brimeyer M.O., Mehrota A., Quici S., Nigam A., Regen S.L.
Journal of Organic Chemistry,
1980
21.

Akhlaghinia B., Rahimizadeh M., Eshghi H., Zhaleh S., Rezazadeh S.
Journal of Sulfur Chemistry,
2012
22.

Wannagat U., Damrath V., Schliephake A., Harder U.
Monatshefte fur Chemie,
1987
23.

Jones R.A., Katritzky A.R., Lehman P.G., Richards A.C., Scattergood R.
Journal of the Chemical Society Perkin Transactions 2,
1972
24.

Bouda H., Borredon M.E., Delmas M., Gaset A.
Synthetic Communications,
1987
25.
![A highly active two six-membered phosphinite palladium PCP pincer complex [PdCl{C6H3(CH2OPPri)2-2,6}]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Naghipour A., Sabounchei S.J., Morales-Morales D., Canseco-González D., Jensen C.M.
Polyhedron,
2007
26.
![SPS–Ni(II) pincer compounds of the type [Ni(phPS2)(P(C6H4-4-R)3)] Synthesis, characterization and catalytic evaluation in C S cross-coupling reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gómez-Benítez V., Valdés H., Hernández-Ortega S., Manuel German-Acacio J., Morales-Morales D.
Polyhedron,
2018
27.

López-Saucedo F., Flores-Rojas G.G., González-Sebastián L., Reyes-Martínez R., German-Acacio J.M., Avila-Sorrosa A., Hernández-Ortega S., Morales-Morales D.
Inorganica Chimica Acta,
2018
28.

Valdés H., García-Eleno M.A., Canseco-Gonzalez D., Morales-Morales D.
ChemCatChem,
2018
29.
10.1016/j.mencom.2021.11.026_b0145
González-Sebastián
D,
2018
30.
10.1016/j.mencom.2021.11.026_b0150
Flores-Rojas
Polyhedron,
2020
31.

Koning B., Hulst R., Bouter A., Buter J., Meetsma A., Kellogg R.M.
Chemical Communications,
1997
32.

Steiner T.
Angewandte Chemie - International Edition,
2002
33.

Haouas M., Taulelle F., Martineau C.
Progress in Nuclear Magnetic Resonance Spectroscopy,
2016