Keywords
acid–base reaction
excited state
fluorescence
micelles
photobase
photoinduced proton transfer
protonation
shift in equilibrium
surfactants
Abstract
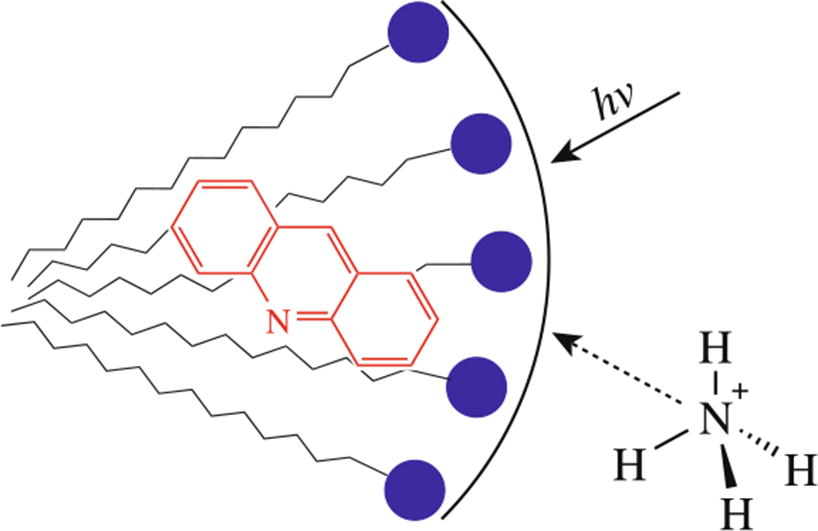
The effect of anionic, nonionic and cationic surfactants on the protonation reaction of acridine in an excited state has been studied. In the case of sodium decyl sulfate and sodium dodecyl sulfate, the observed shift in pK * to a more alkaline region was +0.6 and +0.9 units (from 9.9 to 10.5 and 10.8 respectively), which corresponds to a decrease in the Gibbs free energy by 3.5 and 5.0 kJ mol–1.
References
1.
10.1016/j.mencom.2021.11.021_b0005
Lakowicz
Principles of Fluorescence Spectroscopy,
2006
2.

Wan P., Shukla D.
Chemical Reviews,
1993
3.

Li Y., Feng X., Wang A., Yang Y., Fei J., Sun B., Jia Y., Li J.
Angewandte Chemie - International Edition,
2019
4.

Das A., Ayad S., Hanson K.
Organic Letters,
2016
5.
Sorokin I.D., Gromov O.I., Pergushov V.I., Pomogailo D.A., Melnikov M.Y.
Mendeleev Communications,
2020
6.

Wang C., Fu J., Yao K., Xue K., Xu K., Pang X.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2018
7.

Nunes M.C., dos Santos Carlos F., Fuganti O., Galindo D.D., De Boni L., Abate G., Nunes F.S.
Inorganica Chimica Acta,
2020
8.

Lee S.C., Park S., So H., Lee G., Kim K., Kim C.
Sensors,
2020
9.
Budyka M.F., Biktimirova N.V., Gavrishova T.N., Laukhina O.D.
Mendeleev Communications,
2004
10.

Zhang Z., Zhang H., Yuan X., Yu K.
Industrial & Engineering Chemistry Research,
2020
11.

Selivanov N.I., Samsonova L.G., Solodova T.A., Kopylova T.N., Tel’minov E.N.
Optics and Spectroscopy (English translation of Optika i Spektroskopiya),
2013
12.

Dutta A., Dutta R.K.
Journal of Molecular Liquids,
2013
13.

Totland C., Thomas P.J., Holst B., Akhtar N., Hovdenes J., Skodvin T.
RSC Advances,
2019
14.

Khurana R., Barooah N., Bhasikuttan A.C., Mohanty J.
Organic and Biomolecular Chemistry,
2017
15.
R. Hazarika, R. K. Dutta and S. N. Bhat, Indian J. Chem., Sect. A: Inorg., Bio-inorg., Phys., Theor. Anal. Chem., 1993, 32, 239.
16.

Naumova A.O., Melnikov P.V., Dolganova E.V., Yashtulov N.A., Zaitsev N.K.
Fine Chemical Technologies,
2020
17.
10.1016/j.mencom.2021.11.021_b0085
Mugabutaeva
Ser Mater. Sci. Eng.,
2021
18.

Naumova A.O., Mugabutaeva A.S., Melnikov P.V., Zaitsev N.K.
Moscow University Chemistry Bulletin,
2021
19.
10.1016/j.mencom.2021.11.021_b0095
Liu
Chin. J. Anal. Chem.,
1934
20.

Yankova T.V., Melnikov P.V., Zaitsev N.K.
Moscow University Chemistry Bulletin,
2019
21.

Yankova T.V., Melnikov P.V., Zaytsev N.K.
Moscow University Chemistry Bulletin,
2020
22.

Andrade S.M., Costa S.M.
Photochemical and Photobiological Sciences,
2002
23.

Falcone R.D., Correa N.M., Biasutti M.A., Silber J.J.
Langmuir,
2002
24.

Shaw A.K., Pal S.K.
Journal of Physical Chemistry B,
2007
25.

Kamanin S.S., Arlyapov V.A., Machulin A.V., Alferov V.A., Reshetilov A.N.
Russian Journal of Applied Chemistry,
2015
26.

Kamanina O.A., Kamanin S.S., Kharkova A.S., Arlyapov V.A.
3 Biotech,
2019
27.

Wolfbeis O.S.
BioEssays,
2015
28.
10.1016/j.mencom.2021.11.021_b0140
Ryan
J. Phys. Chem. A,
1827
29.
10.1016/j.mencom.2021.11.021_b0145
Dickerson
Chemistry, Matter, and the Universe: An Integrated Approach to General Chemistry,
1976
30.

Lakowicz J.R., Balter A.
Biophysical Chemistry,
1982
31.

Amado A.M., Ramos A.P., Silva E.R., Borissevitch I.E.
Journal of Luminescence,
2016
32.

Kellmann A.
The Journal of Physical Chemistry,
1977
33.

Bowen E.J., Holder N.J., Woodger G.B.
The Journal of Physical Chemistry,
1962