Keywords
controlled drug delivery
cytotoxicity
molecular switch
pH-sensitive liposome
stimulus-sensitive
Abstract
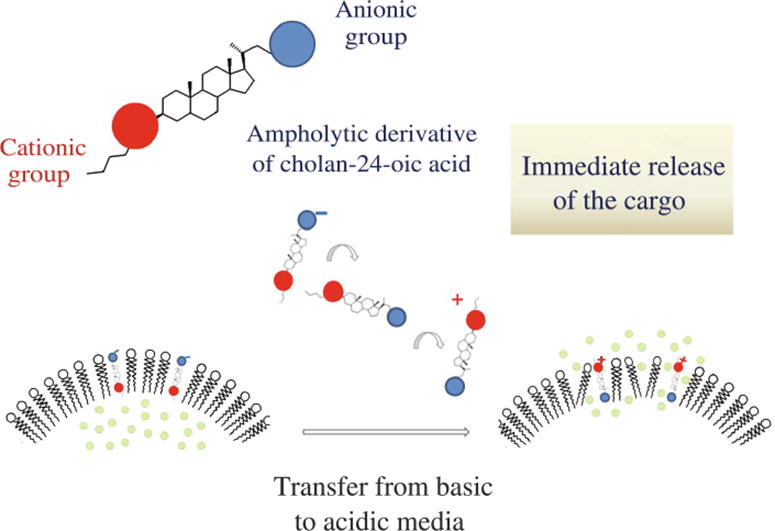
Stimulus-sensitive liposomes have been prepared from zwitterionic dioleoylphosphocholine and ampholytic molecular switches with carboxylic anionic groups and the triazole or isobutylamino cationic ones attached to the opposite ends of the steroid core. When the pH of outer solution was altered from slightly alkaline to slightly acidic, the switches changed their orientation in the liposomal membrane, which induced temporal defects formation and the release of a drug model load. The low-toxic pH-sensitive isobutylamino derivative–dioleoylphosphocholine liposomes demonstrated fast cargo release.
References
1.

Lehner R., Wang X., Marsch S., Hunziker P.
Nanomedicine: Nanotechnology, Biology, and Medicine,
2013
2.

Allen T.M., Cullis P.R.
Advanced Drug Delivery Reviews,
2013
3.
10.1016/j.mencom.2021.11.019_b0015
Lao
J. Drug Delivery,
2013
4.
Denieva Z.G., Budanova U.A., Sebyakin Y.L.
Mendeleev Communications,
2019
5.

Bozzuto G., Molinari A.
International Journal of Nanomedicine,
2015
6.

Kheirolomoom A., Lai C., Tam S.M., Mahakian L.M., Ingham E.S., Watson K.D., Ferrara K.W.
Journal of Controlled Release,
2013
7.
10.1016/j.mencom.2021.11.019_b0040
Helmlinger
Clin. Cancer Res.,
2002
8.

Kato Y., Ozawa S., Miyamoto C., Maehata Y., Suzuki A., Maeda T., Baba Y.
Cancer Cell International,
2013
9.

Torchilin V.P.
Annual Review of Biomedical Engineering,
2006
10.

Aryasomayajula B., Salzano G., Torchilin V.P.
Methods in Molecular Biology,
2017
11.

Monteiro L.O., Malachias Â., Pound-Lana G., Magalhães-Paniago R., Mosqueira V.C., Oliveira M.C., de Barros A.L., Leite E.A.
Langmuir,
2018
12.
Tokudome Y., Nakamura K., Itaya Y., Hashimoto F.
Journal of Pharmacy and Pharmaceutical Sciences,
2015
13.

Paliwal S.R., Paliwal R., Pal H.C., Saxena A.K., Sharma P.R., Gupta P.N., Agrawal G.P., Vyas S.P.
Molecular Pharmaceutics,
2011
14.

Ferreira D.D., Lopes S.C., Franco M.S., Oliveira M.C.
Therapeutic Delivery,
2013
15.

Paliwal S.R., Paliwal R., Vyas S.P.
Drug Delivery,
2014
16.

Guo X., Gagne L., Chen H., Szoka F.C.
Journal of Liposome Research,
2013
17.

Viricel W., Mbarek A., Leblond J.
Angewandte Chemie - International Edition,
2015
18.

Zheng Y., Liu X., Samoshina N.M., Samoshin V.V., Franz A.H., Guo X.
Chemistry and Physics of Lipids,
2018
19.

Samoshina N.M., Liu X., Brazdova B., Franz A.H., Samoshin V.V., Guo X.
Pharmaceutics,
2011
20.

Yaroslavov A., Efimova A., Smirnova N., Erzunov D., Lukashev N., Grozdova I., Melik-Nubarov N.
Colloids and Surfaces B: Biointerfaces,
2020
21.
Efimova A.A., Kostenko S.N., Orlov V.N., Yaroslavov A.A.
Mendeleev Communications,
2016
22.
Yaroslavov A.A., Efimova A.A., Mulashkin F.D., Rudenskaya G.N., Krivtsov G.G.
Mendeleev Communications,
2018
23.

Efimova A.A., Mulashkin F.D., Rudenskaya G.N., Evtushenko E.G., Orlov V.N., Melik-Nubarov N.S., Krivtsov G.G., Yaroslavov A.A.
Polymer Science - Series B,
2018