Keywords
aldoximes
arylnitromethanes
hydrogen peroxide
oxidation
perborate
urea
Abstract
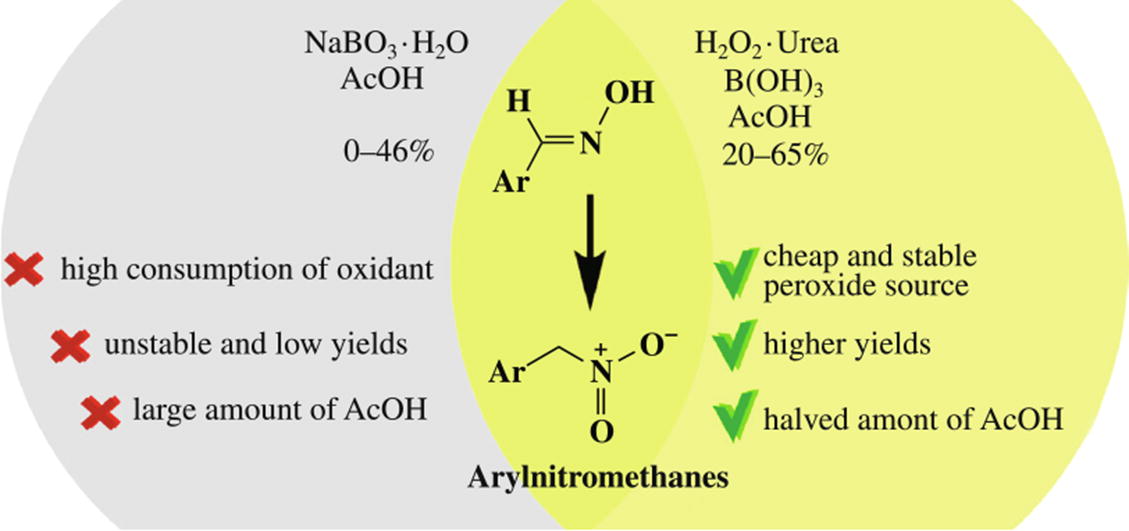
Arylnitromethanes were prepared by oxidation of aldoximes with sodium perborate or with urea hydrogen peroxide in the presence of boric acid. The use of urea hydrogen peroxide complex provides better and more stable yields of nitro compound and allows one to halve an acetic acid consumption. Only E-aldoximes are oxidized to arylnitromethanes while Z-isomers are transformed into complex mixtures not containing arylnitromethanes.
References
1.

N. V. G. M., Dyapa R., Pansare S.V.
Organic Letters,
2015
2.

Gonzalez A.Z., Eksterowicz J., Bartberger M.D., Beck H.P., Canon J., Chen A., Chow D., Duquette J., Fox B.M., Fu J., Huang X., Houze J.B., Jin L., Li Y., Li Z., et. al.
Journal of Medicinal Chemistry,
2014
3.
(a) F. Nietzold, S. Rubner and T. Berg, Chem. Commun., 2019, 55, Asian J., 2015, 10, 334.
4.

Silyanova E.A., Samet A.V., Salamandra L.K., Khrustalev V.N., Semenov V.V.
European Journal of Organic Chemistry,
2020
5.

Semenova M.N., Demchuk D.V., Tsyganov D.V., Chernysheva N.B., Samet A.V., Silyanova E.A., Kislyi V.P., Maksimenko A.S., Varakutin A.E., Konyushkin L.D., Raihstat M.M., Kiselyov A.S., Semenov V.V.
ACS Combinatorial Science,
2018
6.

Chernysheva N.B., Maksimenko A.S., Andreyanov F.A., Kislyi V.P., Strelenko Y.A., Khrustalev V.N., Semenova M.N., Semenov V.V.
European Journal of Medicinal Chemistry,
2018
7.
10.1016/j.mencom.2021.11.016_b0025
Ono
The Nitro Group in Organic Synthesis, ed,
2001
8.
(a) G. Occhipinti, L. Liguori, A. Tsoukala and H. R. Bjorsvik, Org.
9.

Occhipinti G., Liguori L., Tsoukala A., Bjørsvik H.
Organic Process Research and Development,
2010
10.

Nishiwaki Y., Sakaguchi S., Ishii Y.
Journal of Organic Chemistry,
2002
11.

Suzuki H., Murashima T., Kozai I., Mori T.
Journal of the Chemical Society Perkin Transactions 1,
1993
12.

Kornblum N., Smiley R.A., Blackwood R.K., Iffland D.C.
Journal of the American Chemical Society,
1955
13.

Hauser F.M., Baghdanov V.M.
Journal of Organic Chemistry,
1988
14.

Sedelmeier J., Ley S.V., Baxendale I.R., Baumann M.
Organic Letters,
2010
15.

Freeman F., Yeramyan A.
Journal of Organic Chemistry,
1970
16.

Walvoord R.R., Berritt S., Kozlowski M.C.
Organic Letters,
2012
17.

Jiang M., Feng L., Feng J., Jiao P.
Organic Letters,
2017
18.

Matviitsuk A., Greenhalgh M.D., Taylor J.E., Nguyen X.B., Cordes D.B., Slawin A.M., Lupton D.W., Smith A.D.
Organic Letters,
2019
19.

Akulov A.A., Varaksin M.V., Charushin V.N., Chupakhin O.N.
Russian Chemical Reviews,
2021
20.

Ballini R., Marcantoni E., Petrini M.
Tetrahedron Letters,
1992
21.

Goti A., Cardona F., Soldaini G.
Synlett,
2004
22.

Tsukanov S.V., Johnson M.D., May S.A., Rosemeyer M., Watkins M.A., Kolis S.P., Yates M.H., Johnston J.N.
Organic Process Research and Development,
2016
23.

Vara B.A., Mayasundari A., Tellis J.C., Danneman M.W., Arredondo V., Davis T.A., Min J., Finch K., Guy R.K., Johnston J.N.
Journal of Organic Chemistry,
2014
24.

Base D.S., Vanajatha G.
Synthetic Communications,
1998
25.

Tyman J.H., Payne P.B.
Journal of Chemical Research,
2006
26.

Tsukanov S.V., Johnson M.D., May S.A., Kolis S.P., Yates M.H., Johnston J.N.
Organic Process Research and Development,
2018
27.

Olah G.A., Ramaiah P., Lee C., Surya Prakash G.K.
Synlett,
1992
28.

Emmons W.D., Pagano A.S.
Journal of the American Chemical Society,
1955
29.

McKillop A., Sanderson W.R.
Journal of the Chemical Society Perkin Transactions 1,
2000
30.
(a) M. A. A. F. de C. T. Carrondo and A. C. Skapski, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1978, 34, 3551
31.

Onak T., Landesman H., Williams R., Shapiro I.
The Journal of Physical Chemistry,
1959
32.

Flanagan J., Griffith W.P., Powell R.D., West A.P.
Journal of the Chemical Society Dalton Transactions,
1989
33.
(d) B. N. Chernyshov, O. V. Brovkina, B. Ya. Kavun and E. B. Pashnina, Russ. J. Inorg. Chem., 1996, 41, 1705 (Zh. Neorg. Khim., 1996, 41, 1798). 13 M. S. Manna and S. Mukherjee, J. Am. Chem. Soc., 2015, 137, 130.
34.

Lobachev V.L., Dyatlenko L.M., Zubritskii M.Y.
Kinetics and Catalysis,
2016
35.

Vishnuvarthan M., James Paterson A., Raja R., Piovano A., Bonino F., Gianotti E., Berlier G.
Microporous and Mesoporous Materials,
2011
36.

Raja R., Thomas J.M., Greenhill-Hooper M., Doukova V.
Chemical Communications,
2007
37.
10.1016/j.mencom.2021.11.016_b0075
Pinnick
Org. React.,
1990
38.
10.1016/j.mencom.2021.11.016_b0080
Gawley
Org. React.,
1988
39.

Blackwell M., Dunn P.J., Graham A.B., Grigg R., Higginson P., Saba I.S., Thornton-Pett M.
Tetrahedron,
2002