Keywords
copper catalysis
cyanation
diazonium salts
Meerwein arylation
photocatalysis
styrenes
Abstract
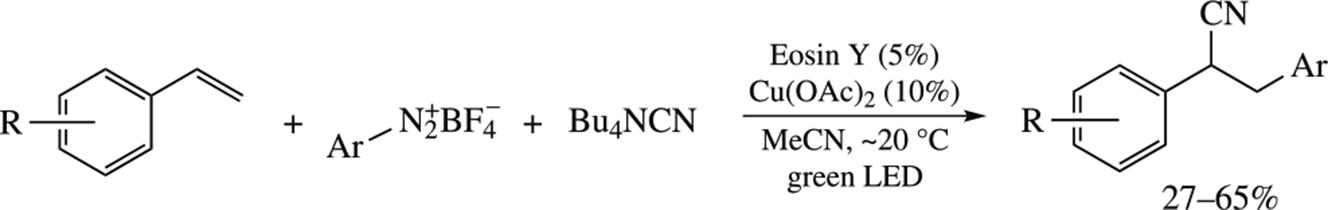
A new procedure for the cyanoarylation of styrenes with arenediazonium salts and Bu4NCN employs double catalytic system Eosyn Y/Cu(OAc)2 under the irradiation with green light (530 nm). In the case of diazonium salts bearing electron withdrawing substituents, the products are formed in 40–65% yields. The relative thiocyanatoarylation proceeds only in the presence of photocatalyst, but the Cu(OAc)2 additive allows for the product yield improvement
References
1.

Heinrich M., Kindt S.
Synthesis,
2016
2.

Mo F., Qiu D., Zhang L., Wang J.
Chemical Reviews,
2021
3.

Dombrovskii A.V.
Russian Chemical Reviews,
1984
4.

Hari D.P., König B.
Angewandte Chemie - International Edition,
2013
5.

Zhu R., Buchwald S.L.
Journal of the American Chemical Society,
2015
6.

Baranovskii V.S., Petrushka B.M., Fesak A.Y., Grishchuk B.D.
Russian Journal of General Chemistry,
2013
7.

Xiao Z., Liu Y., Zheng L., Liu C., Guo Y., Chen Q.
Journal of Organic Chemistry,
2018
8.

Kindt S., Wicht K., Heinrich M.R.
Angewandte Chemie - International Edition,
2016
9.

de Souza E.L., Wiethan C., Correia C.R.
ACS Omega,
2019
10.

Obushak N.D., Matiichuk V.S., Martyak R.L., Ganushchak N.I.
Chemistry of Heterocyclic Compounds,
1999
11.

Batsyts S., Shehedyn M., Goreshnik E.A., Obushak M.D., Schmidt A., Ostapiuk Y.V.
European Journal of Organic Chemistry,
2019
12.

Fedorov O.V., Scherbinina S.I., Levin V.V., Dilman A.D.
Journal of Organic Chemistry,
2019
13.

Chen X., Gui Q., Yi R., Yu X., Wu Z., Huang Y., Cao Z., He W.
Organic and Biomolecular Chemistry,
2020
14.
10.1016/j.mencom.2021.11.015_b0050
Obushak
Russ. J. Org. Chem.,
1996
15.

Zhuang W., Chen P., Liu G.
Chinese Journal of Chemistry,
2020
16.

Wicholas M., Wolford T.
Inorganic Chemistry,
1974
17.

Yamaguchi E., Tanaka W., Itoh A.
Chemistry - An Asian Journal,
2018
18.

Hering T., Hari D.P., König B.
Journal of Organic Chemistry,
2012
19.

Hoque I.U., Chowdhury S.R., Maity S.
Journal of Organic Chemistry,
2019
20.

Prasad Hari D., Hering T., König B.
Angewandte Chemie - International Edition,
2013
21.

Yao C., Sun Q., Rastogi N., König B.
ACS Catalysis,
2015
22.

Ouyang X., Cheng J., Li J.
Chemical Communications,
2018
23.

Anselmo M., Moni L., Ismail H., Comoretto D., Riva R., Basso A.
Beilstein Journal of Organic Chemistry,
2017
24.
![Visible-light-induced photoredox-catalyzed synthesis of benzimidazo[2,1-a]iso-quinoline-6(5H)-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wang B., Zou L., Wang L., Sun M., Li P.
Chinese Chemical Letters,
2021
25.

Zlotin S.G., Dalinger I.L., Makhova N.N., Tartakovsky V.A.
Russian Chemical Reviews,
2020