Keywords
cyanine dyes
dyads
fluorescence
fullerene C60
fullerene derivatives
heptamethine compounds
NIR absorbance
Abstract
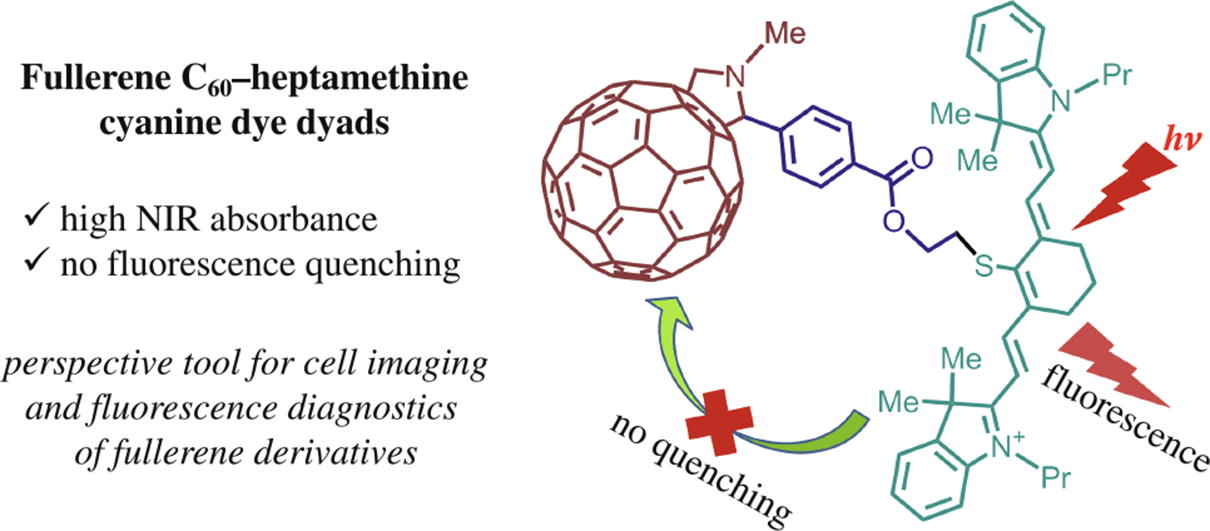
Two new heptamethine cyanine dye–fullerene C60 covalently- linked dyads, which absorb in far-red and NIR spectral regions, have been synthesized by esterification click reaction and characterized by physicochemical methods. No significant fluorescence quenching was found due to weak electronic coupling between heptamethine moiety and fullerene core, which was confirmed by photophysical and electrochemical methods. Such dyads can be useful for cell imaging and fluorescence diagnostics of various fullerene derivatives.
References
1.

Karmova F.M., Lebedeva V.S., Mironov A.F.
Russian Journal of General Chemistry,
2016
2.

Zhao J., Wu W., Sun J., Guo S.
Chemical Society Reviews,
2013
3.

Belik A.Y., Rybkin A.Y., Voronov I.I., Goryachev N.S., Volyniuk D., Grazulevicius J.V., Troshin P.A., Kotelnikov A.I.
Dyes and Pigments,
2017
4.

Rybkin A.Y., Belik A.Y., Kraevaya O.A., Khakina E.A., Zhilenkov A.V., Goryachev N.S., Volyniuk D., Grazulevicius J.V., Troshin P.A., Kotelnikov A.I.
Dyes and Pigments,
2019
5.
![Efficient light harvesting anionic heptamethine cyanine–[60] and [70]fullerene hybrids](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Villegas C., Krokos E., Bouit P., Delgado J.L., Guldi D.M., Martín N.
Energy and Environmental Science,
2011
6.

Bouit P., Spänig F., Kuzmanich G., Krokos E., Oelsner C., Garcia-Garibay M., Delgado J., Martín N., Guldi D.
Chemistry - A European Journal,
2010
7.

Rybkin A.Y., Belik A.Y., Tarakanov P.A., Taziev K.R., Kozlov A.V., Goryachev N.S., Sulimenkov I.V., Kozlovskiy V.I., Romanenko Y.V., Koifman O.I., Kotelnikov A.I.
Macroheterocycles,
2019
8.

Ma X., Laramie M., Henary M.
Bioorganic and Medicinal Chemistry Letters,
2018
9.

Levitz A., Marmarchi F., Henary M.
Molecules,
2018
10.
K. Rurack, in Standardization and Quality Assurance in Fluorescence Measurements I: Techniques, ed. U. Resch-Genger, Springer, Berlin, Heidelberg, 2008, pp. 101–145
11.

Kozlov A.V., Rybkin A.Y., Belik A.Y., Taziev K.R., Tarakanov P.A., Goryachev N.S., Sulimenkov I.V., Kozlovskiy V.I., Romanenko Y.V., Koifman M.O., Gostev F.E., Shelaev I.V., Aybush A.V., Nadtochenko V.A., Kotelnikov A.I., et. al.
Macroheterocycles,
2020
12.
![Self-assembling nanostructures of water-soluble fullerene[60]–chlorin e6 dyads: Synthesis, photophysical properties, and photodynamic activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Rybkin A.Y., Belik A.Y., Goryachev N.S., Mikhaylov P.A., Kraevaya O.A., Filatova N.V., Parkhomenko I.I., Peregudov A.S., Terent'ev A.A., Larkina E.A., Mironov A.F., Troshin P.A., Kotelnikov A.I.
Dyes and Pigments,
2020
13.
![N-phenyl[60]fulleropyrrolidines: alternative acceptor materials to PC61BM for high performance organic photovoltaic cells](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Karakawa M., Nagai T., Adachi K., Ie Y., Aso Y.
Journal of Materials Chemistry A,
2014
14.

Chernyak A.V., Avilova I.A., Khakina E.A., Mumyatov A.V., Zabrodin V.A., Troshin P.A., Volkov V.I.
Applied Magnetic Resonance,
2016
15.
Tuktarov A.R., Khuzin A.A., Dzhemilev U.M.
Mendeleev Communications,
2019
16.
![[60]Fullerene as a Substituent](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bagno A., Claeson S., Maggini M., Martini M.L., Prato M., Scorrano G.
Chemistry - A European Journal,
2002