Keywords
N-nitrooxazolidine
N-nitroperhydro-1,3-oxazine
azidation
azides
chlorination
nitramine
phase transfer catalyst
thionyl chloride
α,μ-dichloro-2-nitro- 2-azaalkanes
Abstract
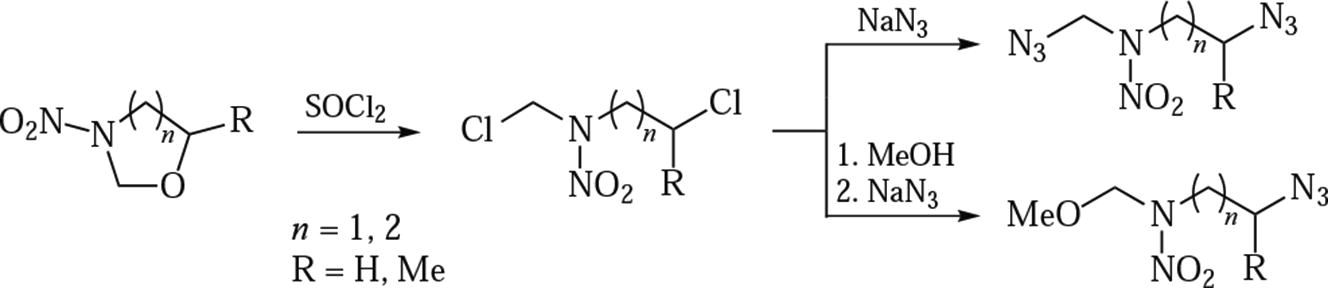
A new versatile access to azido-substituted N-alkylnitramines is based on ring opening in N-nitrooxazolidines and N-nitroperhydro-1,3-oxazines with thionyl chloride followed by treatment with sodium azide. Representative N,N-bis(azidoalkyl)- and N-azidoalkyl-N-methoxymethyl- containing nitramines were synthesized and characterized.
References
1.
V. M. Zinov’ev, G. V. Kutsenko and A. S. Ermilov, Sovremennye I perspektivnye vysokoenergeticheskie komponenty smesevykh I ballisticheskikh tverdykh raketnykh topliv (Modern and Prospective High-Energetic Components of Composite and Ballistic Solid Propellants), PGTU publishing, Perm, 2010 (in Russian).
2.
10.1016/j.mencom.2021.11.008_b0010
Simmons
25th International Annual Conference of ICT ‘Thermochemistry of NENA Plasticizers’,
1994
3.
V. P. Ivshin, M. S. Komelin and N. P. Belik, J. Org. Chem. USSR, 1980, 16, 1006 (Zh. Org. Khim., 1980, 16, 1166).
4.
G. A. Marchenko, L. N. Punegova, T. S. Shitova, N. A. Roman’ko, L. S. Egorova and Yu. M. Kargin, J. Org. Chem. USSR, 1986, 22, 33 (Zh. Org. Khim., 1986, 22, 40).
5.

Tartakovsky V.A., Ermakov A.S., Strelenko Y.A., Vinogradov D.B., Serkov S.A.
Russian Chemical Bulletin,
2001
6.

Zlotin S.G., Arabadzhi S.S., Zharkov M.N., Kuchurov I.V.
Synthesis,
2020
7.

Witucki E.F., Wilson E.R., Flanagan J.E., Frankel M.B.
Journal of Chemical & Engineering Data,
1983
8.

Unterhalt B., Leiblein F.
Archiv der Pharmazie,
1978