Keywords
azoxy compounds
combustion calorimetry
differential scanning calorimetry
enthalpy of formation
furazans
nucleophilic substitution
X-ray diffraction analysis
Abstract
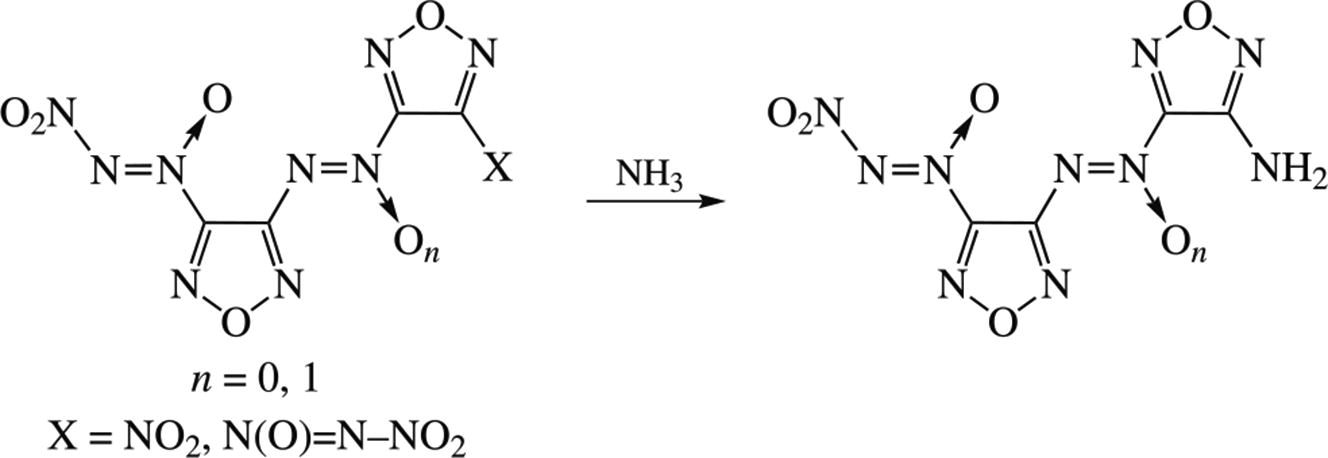
Novel energetic azoxy- and azofurazans bearing nitro-NNO- azoxy and amino groups were synthesized using the ammonolysis of some known (nitro-NNO-azoxy)furazans. 3-Amino-4-{[4′-(nitro-NNO-azoxy)f urazan-3′-yl]-NNO- azoxy}furazan displays the highest melting point (114 °C, decomp.) among the known (nitro-NNO-azoxy)furazans, optimal density (1.80 g cm−3), high experimental enthalpy of formation (639 kcal kg−1) and mechanical sensitivity on the level of PETN. In terms of the specific impulse level, the model solid composite propellant formulations based on this compound outperform similar formulations based on RDX, HMX or CL-20 by 7–12 s.
References
1.
10.1016/j.mencom.2021.11.006_b0005
Lempert
Cent. Eur. J. Energ. Mater.,
2011
2.
10.1016/j.mencom.2021.11.006_b0010
Lempert
Chin. J. Explos. Propellants,
2015
3.
10.1016/j.mencom.2021.11.006_b0015
Klapötke
Chemistry of High-Energy Materials,
2017
4.

O’Sullivan O.T., Zdilla M.J.
Chemical Reviews,
2020
5.

Gao H., Zhang Q., Shreeve J.M.
Journal of Materials Chemistry A,
2020
6.

Chavez D.E.
Topics in Heterocyclic Chemistry,
2017
7.
10.1016/j.mencom.2021.11.006_b0035
Agrawal
Organic Chemistry of Explosives,
2007
8.

Sheremetev A.B., Semenov S.E., Kuzmin V.S., Strelenko Y.A., Ioffe S.L.
Chemistry - A European Journal,
1998
9.

Wang Y., Li S., Li Y., Zhang R., Wang D., Pang S.
Journal of Materials Chemistry A,
2014
10.

Li H., Wang B.Z., Li X.Z., Tong J.F., Lai W.P., Fan X.Z.
Bulletin of the Korean Chemical Society,
2013
11.
10.1016/j.mencom.2021.11.006_b0055
Li
Chin. J. Energ. Mater.,
2014
12.

Lai W., Lian P., Liu Y., Yu T., Zhu W., Ge Z., Lv J.
Journal of Molecular Modeling,
2014
13.
10.1016/j.mencom.2021.11.006_b0065
Lian
Chin. J. Explos. Propellants,
2014
14.

Leonov N.E., Klenov M.S., Anikin O.V., Churakov A.M., Strelenko Y.A., Voronin A.A., Lempert D.B., Muravyev N.V., Fedyanin I.V., Semenov S.E., Tartakovsky V.A.
ChemistrySelect,
2020
15.
M. S. Klenov, N. E. Leonov, A. A. Guskov, A. M. Churakov, Yu. A. Strelenko and V. A. Tartakovsky, Russ. Chem. Bull., Int. Ed., 2019, 68, 1798 (Izv. Akad. Nauk, Ser. Khim., 2019, 1798).
16.

Anikin O.V., Leonov N.E., Klenov M.S., Churakov A.M., Voronin A.A., Muravyev N.V., Strelenko Y.A., Fedyanin I.V., Tartakovsky V.A.
European Journal of Organic Chemistry,
2019
17.

Leonov N.E., Klenov M.S., Anikin O.V., Churakov A.M., Strelenko Y.A., Monogarov K.A., Tartakovsky V.A.
European Journal of Organic Chemistry,
2018
18.
Churakov A.M., Ioffe S.L., Tartakovskii V.A.
Mendeleev Communications,
1996
19.

Churakov A.M., Semenov S.E., Ioffe S.L., Strelenko Y.A., Tartakovsky V.A.
Russian Chemical Bulletin,
1997
20.
10.1016/j.mencom.2021.11.006_b0100
High Energy Density Materials,
2007
21.
10.1016/j.mencom.2021.11.006_b0105
Zhilin
Malochuvstvitel’nye vzryvchatye veshchestva (Insensitive Explosives),
2008
22.

Sheremetev A.B., Kulagina V.O., Kryazhevskikh I.A., Melnikova T.M., Aleksandrova N.S.
Russian Chemical Bulletin,
2002
23.
A. B. Sheremetev, O. V. Kharitonova, E. V. Mantseva, V. O. Kulagina, E. V. Shatunova, N. S. Aleksandrova, T. M. Mel’nikova, E. A. Ivanova, D. E. Dmitriev, V. A. Eman, I. L. Yudin, V. S. Kuz’min, Yu. A. Strelenko, T. S. Novikova, O. V. Lebedev and L. I. Khmel’nitskii, Russ. J. Org.Chem., 1999, 35, 1525 (Zh. Org. Khim., 1999, 35, 1555).
24.

Dmitriev D.E., Strelenko Y.A., Sheremetev A.B.
Russian Chemical Bulletin,
2013
25.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
26.

Luk′yanov O.A., Parakhin V.V., Shlykova N.I., Dmitrienko A.O., Melnikova E.K., Kon'kova T.S., Monogarov K.A., Meerov D.B.
New Journal of Chemistry,
2020
27.
10.1016/j.mencom.2021.11.006_b0135
Sumin
Transactions of the 11th International Detonation Symposium,
1998
28.
10.1016/j.mencom.2021.11.006_b0140
Meyer
Explosives,
2016