Keywords
ammonium
bis(furazano)pyrazine
McConnell’s exchange model
molecular magnet
potassium
rubidium
sodium
stable radical
Abstract
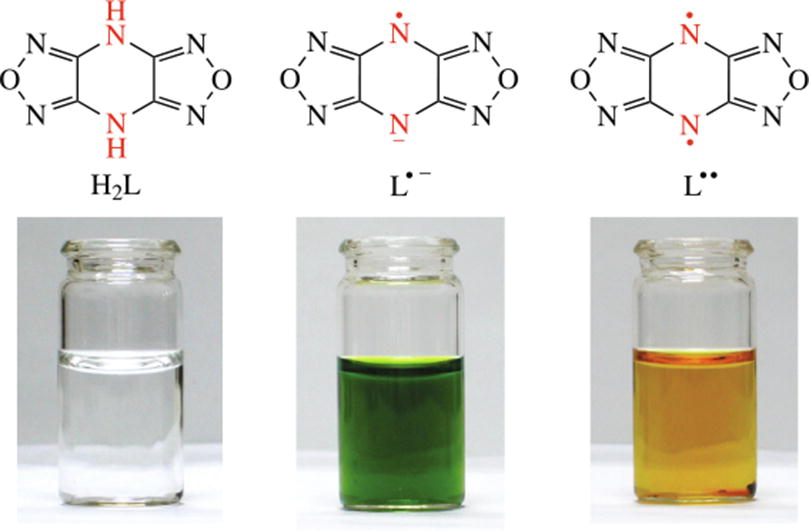
The first organic magnet based on a high-nitrogen framework of pyrazine-fused furazans Na(L•−)(H2O)3 was found. A quantum-chemical study of M(L•−)(H2O)n, where M = Li, Na, K, Rb, NH4, revealed that exchange coupling energy between the neighboring radical anions proved highly sensitive to the motion of one L•− relative to another.
References
1.
10.1016/j.mencom.2021.11.005_b0005
Organic Chemistry of Stable Free Radicals,
1968
2.
Volodarsky L.B., Reznikov V.A., Ovcharenko V.I.
2017
3.
H. Robin, Stable Radicals: Fundamentals and Applied Aspects of Odd-Electron Compounds, ed. R. G. Hicks, John Wiley & Sons, Ltd., Chichester, 2010.
4.

Likhtenshtein G.I.
Springer Series in Materials Science,
2020
5.

Keana J.F.
Chemical Reviews,
1978
6.
10.1016/j.mencom.2021.11.005_b0030
Datta
Theoretical and Computational Aspects of Magnetic Organic Molecules,
2014
7.

Kim Y., Byeon J.E., Jeong G.Y., Kim S.S., Song H., Lee E.
Journal of the American Chemical Society,
2021
8.

Tretyakov E.V., Ovcharenko V.I.
Russian Chemical Reviews,
2009
9.

Suzuki S., Okada K.
2015
10.

Lemaire M.T.
Pure and Applied Chemistry,
2010
11.

12.
10.1016/j.mencom.2021.11.005_b0060
World Scientific Reference on Spin in Organics,
2018
13.

14.
K. Inoue, in π-Electron Magnetism: From Molecules to Magnetic Materials, ed. J. Veciana, Springer, Berlin, Heidelberg, 2001, pp. 61–91
15.

Fidan I., Önal E., Yerli Y., Luneau D., Ahsen V., Hirel C.
ChemPlusChem,
2017
16.
Organic Redox Systems: Synthesis, Properties, and Applications, ed. T. Nishinaga, Wiley, Hoboken, 2016.
17.

18.

19.

Coronado E.
Nature Reviews Materials,
2019
20.

Han W., Pi K., McCreary K.M., Li Y., Wong J.J., Swartz A.G., Kawakami R.K.
Physical Review Letters,
2010
21.

Yonekuta Y., Susuki K., Oyaizu K., Honda K., Hiroyuki Nishide*
Journal of the American Chemical Society,
2007
22.

Oyaizu K., Nishide H.
Advanced Materials,
2009
23.

Lee J., Lee E., Kim S., Bang G.S., Shultz D.A., Schmidt R.D., Forbes M.D., Lee H.
Angewandte Chemie - International Edition,
2011
24.

Gaudenzi R., de Bruijckere J., Reta D., Moreira I.D., Rovira C., Veciana J., van der Zant H.S., Burzurí E.
ACS Nano,
2017
25.

Gaudenzi R., Burzurí E., Reta D., Moreira I.D., Bromley S.T., Rovira C., Veciana J., van der Zant H.S.
Nano Letters,
2016
26.

Herrmann C., Solomon G.C., Ratner M.A.
Journal of the American Chemical Society,
2010
27.
10.1016/j.mencom.2021.11.005_b0135
Hu
Beilstein J. Nanotechnol.,
1919
28.

Tsuji Y., Hoffmann R., Strange M., Solomon G.C.
Proceedings of the National Academy of Sciences of the United States of America,
2016
29.

Gallagher N., Zhang H., Junghoefer T., Giangrisostomi E., Ovsyannikov R., Pink M., Rajca S., Casu M.B., Rajca A.
Journal of the American Chemical Society,
2019
30.

31.

Zhou H., Mayorga-Martinez C.C., Pané S., Zhang L., Pumera M.
Chemical Reviews,
2021
32.

Chiarelli R., Novak M.A., Rassat A., Tholence J.L.
Nature,
1993
33.
10.1016/j.mencom.2021.11.005_b0165
Banister
J. Chem. Soc., Chem. Commun.,
1995
34.
Banister A.J., Bricklebank N., Lavender I., Rawson J.M., Gregory C.I., Tanner B.K., Clegg W., Elsegood M.R., Palacio F.
1996
35.

Rawson J.M., Alberola A., Whalley A.
Journal of Materials Chemistry A,
2006
36.

Miller J.S., Epstein A.J., Reiff W.M.
Science,
1988
37.
I. B. Starchenkov and V. G. Andrianov, Chem. Heterocycl. Compd., 1996, 32, 618 (Khim. Geterotsikl. Soedin., 1996, 717).
38.
I. V. Tselinskii, S. F. Mel’nikova, T. V. Romanova, S. V. Pirogov, G. K. Khisamutdinov, T. A. Mratkhuzina, V. L. Korolev, I. Z. Kondyukov, I. S. Abdrakhmanov and S. P. Smirnov, Russ. J. Org. Chem., 1997, 33, 1656.
39.
![Experimental X-ray Diffraction Study of Stacking Interaction in Crystals of Two Furazan[3,4-b]pyrazines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Averkiev B.B., Korlyukov A.A., Antipin M.Y., Sheremetev A.B., Timofeeva T.V.
Crystal Growth and Design,
2014
40.
I. B. Starchenkov, V. G. Andrianov and A. F. Mishnev, Chem. Heterocycl.Compd., 1999, 35, 499 (Khim. Geterotsikl. Soedin., 1999, 564).
41.
![Chemistry of furazano[3,4-b]pyrazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Starchenkov I.B., Andrianov V.G.
Chemistry of Heterocyclic Compounds,
1997
42.

Zhang J., Dharavath S., Mitchell L.A., Parrish D.A., Shreeve J.M.
Journal of the American Chemical Society,
2016
43.
I. B. Starchenkov, V. G. Andrianov and A. F. Mishnev, Chem. Heterocycl. Compd., 1998, 34, 1081 (Khim. Geterotsikl. Soedin., 1998, 1259).
44.

Snyder C.J., Wells L.A., Chavez D.E., Imler G.H., Parrish D.A.
Chemical Communications,
2019
45.

Gao H., Zhang Q., Shreeve J.M.
Journal of Materials Chemistry A,
2020
46.

Snyder C.J., Imler G.H., Chavez D.E., Parrish D.A.
Crystal Growth and Design,
2021
47.
I. B. Starchenkov, V. G. Andrianov and A. F. Mishnev, Chem. Heterocycl. Compd., 1997, 33, 216 (Khim. Geterotsikl. Soedin., 1997, 250).
48.
![2,2-Bis(methoxy-NNO-azoxy)ethyl Derivatives of 4,8-Dihydro-bis-furazano[3,4-b:3′4′-e]pyrazine: The Synthesis and X-ray Investigation](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zyuzin I.N., Suponitsky K.Y., Sheremetev A.B.
Journal of Heterocyclic Chemistry,
2012
49.
![Chemistry of furazano[3,4-b]pyrazines. 2. Nucleophilic substitution of the azido group in furazano[3,4-b]pyrazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Andrianov V.G., Starchenkov I.B., Mishnev A.F.
Chemistry of Heterocyclic Compounds,
1997
50.
Frumkin A.E., Yudin N.V., Suponitsky K.Y., Sheremetev A.B.
Mendeleev Communications,
2018
51.
I. B. Starchenkov and V. G. Andrianov, Chem. Heterocycl. Compd., 1997, 33, 1352 (Khim. Geterotsikl. Soedin., 1997, 1561).
52.
I. B. Starchenkov, V. G. Andrianov and A. F. Mishnev, Chem. Heterocycl. Compd., 1997, 33, 1355 (Khim. Geterotsikl. Soedin., 1997, 1565).
53.
D. B. Lempert, A. I. Kazakov, A. B. Sheremetev, A. G. Gladyshkin, A. V. Nabatova and L. S. Yanovskii, Russ. Chem. Bull., Int. Ed., 2019, 68, 1856 (Izv. Akad. Nauk, Ser. Khim., 2019, 1856).
54.

Liu N., Duan B., Lu X., Zhang Q., Xu M., Mo H., Wang B.
CrystEngComm,
2019
55.
![Weak Ferromagnetic Ordering of the Li+[TCNE]•–(TCNE = Tetracyanoethylene) Organic Magnet with an Interpenetrating Diamondoid Structure](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Her J., Stephens P.W., Davidson R.A., Min K.S., Bagnato J.D., van Schooten K., Boehme C., Miller J.S.
Journal of the American Chemical Society,
2013
56.
10.1016/j.mencom.2021.11.005_b0280
McConnell
J. Chem. Phys.,
1910
57.

Novoa J.J., Deumal M., Jornet-Somoza J.
Chemical Society Reviews,
2011
58.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015