Keywords
aldehydes
amines
carbon monoxide
chiral complexes
diene complexes
Hayashi ligand
Ketones
reductive amination
rhodium complexes
Abstract
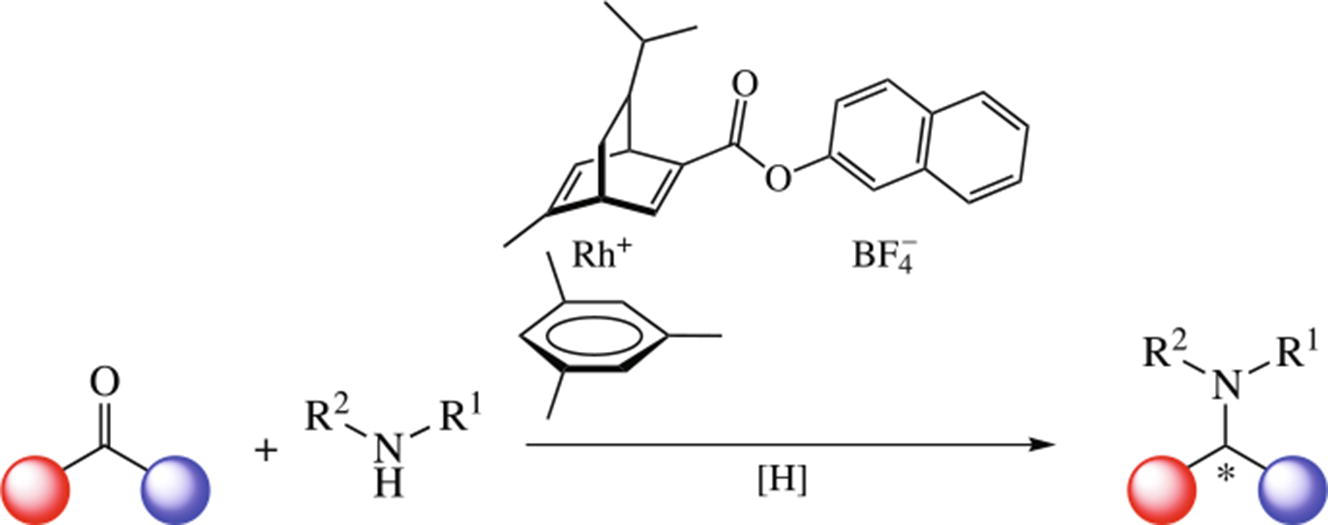
The CO- and H2-assisted reductive amination of carbonyl compounds catalyzed by stable chiral Hayashi ligand-based rhodium complex afforded the racemic amines in moderate yields. The racemic outcome of the process results from the elimination of the chiral ligand from the catalyst under the action of hydrogen or carbon monoxide as reductants.
References
1.

Vatsadze S.Z., Eremina O.E., Veselova I.A., Kalmykov S.N., Nenajdenko V.G.
Russian Chemical Reviews,
2018
2.

Zarezin D.P., Nenajdenko V.G.
Russian Chemical Reviews,
2019
3.

Kovalenko A., Rublev P.O., Tcelykh L.O., Goloveshkin A.S., Lepnev L.S., Burlov A.S., Vashchenko A.A., Marciniak Ł., Magerramov A.M., Shikhaliyev N.G., Vatsadze S.Z., Utochnikova V.V.
Chemistry of Materials,
2019
4.

Katayev D., Larionov E., Nakanishi M., Besnard C., Kündig E.P.
Chemistry - A European Journal,
2014
5.

Wang C., Xiao J.
Topics in Current Chemistry,
2013
6.

Nugent T., El-Shazly M.
Advanced Synthesis and Catalysis,
2010
7.

Patil M.D., Grogan G., Bommarius A., Yun H.
ACS Catalysis,
2018
8.

Li W., Zhang X.
Topics in Current Chemistry,
2013
9.

Sukhorukov A.Y.
Frontiers in Chemistry,
2020
10.

Afanasyev O.I., Kuchuk E., Usanov D.L., Chusov D.
Chemical Reviews,
2019
11.

Patel R.N.
ACS Catalysis,
2011
12.

Ferjancic Z., Saicic R.N.
European Journal of Organic Chemistry,
2021
13.

Dorokhov V.S., Golovanov I.S., Tartakovsky V.A., Sukhorukov A.Y., Ioffe S.L.
Organic and Biomolecular Chemistry,
2018
14.

Sukhorukov A.Y., Dilman A.D., Ioffe S.L.
Chemistry of Heterocyclic Compounds,
2012
15.

Kudryavtsev K.V., Shulga D.A., Chupakhin V.I., Sinauridze E.I., Ataullakhanov F.I., Vatsadze S.Z.
Tetrahedron,
2014
16.

Tian Y., Hu L., Wang Y., Zhang X., Yin Q.
Organic Chemistry Frontiers,
2021
17.

Li C., Villa-Marcos B., Xiao J.
Journal of the American Chemical Society,
2009
18.

Storer R.I., Carrera D.E., Ni Y., MacMillan D.W.
Journal of the American Chemical Society,
2005
19.

Gallardo-Donaire J., Hermsen M., Wysocki J., Ernst M., Rominger F., Trapp O., Hashmi A.S., Schäfer A., Comba P., Schaub T.
Journal of the American Chemical Society,
2017
20.

Mas‐Roselló J., Herraiz A.G., Audic B., Laverny A., Cramer N.
Angewandte Chemie - International Edition,
2020
21.

Janssen-Müller D., Schlepphorst C., Glorius F.
Chemical Society Reviews,
2017
22.

23.

Nakanishi M., Katayev D., Besnard C., Kündig E.P.
Angewandte Chemie - International Edition,
2011
24.

Jia Y., Katayev D., Bernardinelli G., Seidel T., Kündig E. .
Chemistry - A European Journal,
2010
25.

Defieber C., Grützmacher H., Carreira E.
Angewandte Chemie - International Edition,
2008
26.

Nagamoto M., Nishimura T.
ACS Catalysis,
2016
27.

Ankudinov N.M., Chusov D.A., Nelyubina Y.V., Perekalin D.S.
Angewandte Chemie - International Edition,
2021
28.
![C2-Symmetric Bicyclo[2.2.2]octadienes as Chiral Ligands: Their High Performance in Rhodium-Catalyzed Asymmetric Arylation of N-Tosylarylimines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tokunaga N., Otomaru Y., Okamoto K., Ueyama K., Shintani R., Hayashi T.
Journal of the American Chemical Society,
2004
29.

Nishimura T., Yasuhara Y., Hayashi T.
Organic Letters,
2006
30.

Okamoto K., Hayashi T., Rawal V.H.
Organic Letters,
2008
31.
Tsygankov A.A., Makarova M., Chusov D.
Mendeleev Communications,
2018
32.
33.
Afanasyev O.I., Cherkashchenko I., Kuznetsov A., Kliuev F., Semenov S., Chusova O., Denisov G., Chusov D.
Mendeleev Communications,
2020
34.

Dou X., Huang Y., Hayashi T.
Angewandte Chemie - International Edition,
2015
35.

Okamoto K., Hayashi T., Rawal V.H.
Chemical Communications,
2009
36.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Perekalin D.S., Shvydkiy N.V., Maleev V.I., Kudinov A.R., Chusov D.
ACS Catalysis,
2016
37.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Chusov D.
Organic Letters,
2016
38.

Nishimura T., Maeda Y., Hayashi T.
Angewandte Chemie - International Edition,
2010