Keywords
acetic acid
acidity
oxidative carbonylation of methane
water vapor
ZSM-5 zeolites
Abstract
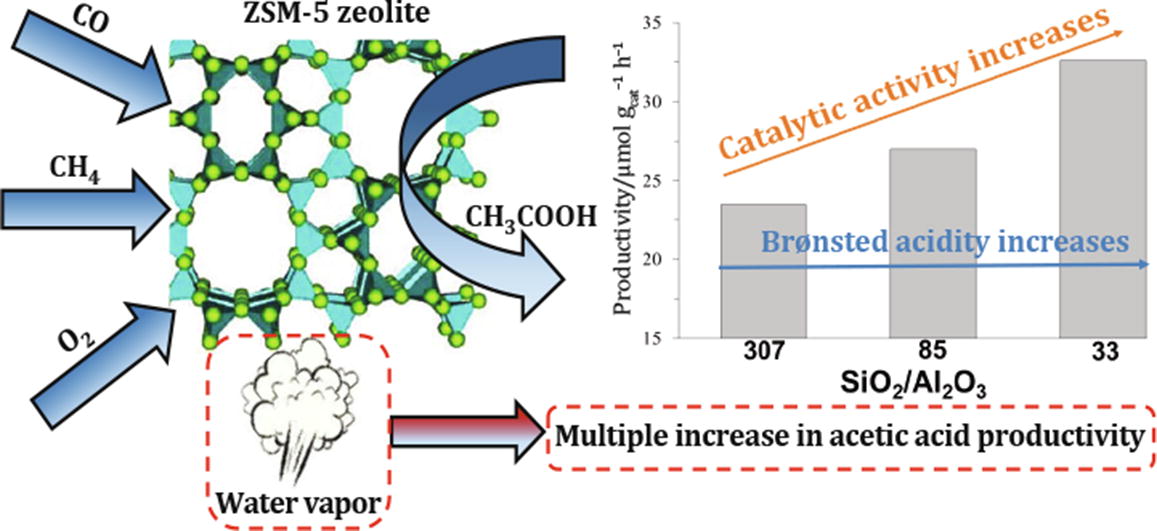
Gas-phase oxidative carbonylation of methane was first performed on ZSM-5 zeolites. The addition of water vapor to a mixture of carbonylation gases leads to a multiple (by two orders of magnitude) increase in acetic acid yield. Zeolites with high acidity, primarily Brønsted acidity, favor the target product formation.
References
1.

Ren Z., Lyu Y., Song X., Ding Y.
Applied Catalysis A: General,
2020
2.

Kalck P., Le Berre C., Serp P.
Coordination Chemistry Reviews,
2020
3.
F. E. Paulik, A. Hershman, W. R. Knox and J. F. Roth, US Patent 3769329, 1973.
4.
B. L. Smith, G. P. Torrence, A. Aguilo’ and J. S. Alder, US Patent 5001259, 1991.
5.

Noriyuki Y., Takeshi M., Joe W., Ben S.
Studies in Surface Science and Catalysis,
1999
6.

Sunley G.J., Watson D.J.
Catalysis Today,
2000
7.

Haynes A., Maitlis P.M., Morris G.E., Sunley G.J., Adams H., Badger P.W., Bowers C.M., Cook D.B., Elliott P.I., Ghaffar T., Green H., Griffin T.R., Payne M., Pearson J.M., Taylor M.J., et. al.
Journal of the American Chemical Society,
2004
8.

Periana R.A., Mironov O., Taube D., Bhalla G., Jones C.
Science,
2003
9.

Phan A., Czaja A.U., Gándara F., Knobler C.B., Yaghi O.M.
Inorganic Chemistry,
2011
10.

YUAN Q., ZHANG Q., WANG Y.
Journal of Catalysis,
2005
11.

Rabie A.M., Betiha M.A., Park S.
Applied Catalysis B: Environmental,
2017
12.

Shavi R., Ko J., Cho A., Han J.W., Seo J.G.
Applied Catalysis B: Environmental,
2018
13.

Tu C., Nie X., Chen J.G.
ACS Catalysis,
2021
14.

Shan J., Li M., Allard L.F., Lee S., Flytzani-Stephanopoulos M.
Nature,
2017
15.

Tang Y., Li Y., Fung V., Jiang D., Huang W., Zhang S., Iwasawa Y., Sakata T., Nguyen L., Zhang X., Frenkel A.I., Tao F.
Nature Communications,
2018
16.
10.1016/j.mencom.2021.09.040_b0080
Wen
Sci. Technol.,
2021
17.

Gunsalus N.J., Koppaka A., Park S.H., Bischof S.M., Hashiguchi B.G., Periana R.A.
Chemical Reviews,
2017
18.
10.1016/j.mencom.2021.09.040_b0090
Zerella
Chem. Commun.,
2004
19.

Chepaikin E.G., Menchikova G.N., Pomogailo S.I.
Russian Chemical Bulletin,
2019
20.

Moteki T., Tominaga N., Ogura M.
ChemCatChem,
2020
21.
10.1016/j.mencom.2021.09.040_b0105
Narsimhan
J. Am. Chem. Soc.,
1825
22.

Bhan A., Iglesia E.
Accounts of Chemical Research,
2008
23.

Vanelderen P., Snyder B.E., Tsai M., Hadt R.G., Vancauwenbergh J., Coussens O., Schoonheydt R.A., Sels B.F., Solomon E.I.
Journal of the American Chemical Society,
2015
24.

Montejo-Valencia B.D., Pagán-Torres Y.J., Martínez-Iñesta M.M., Curet-Arana M.C.
ACS Catalysis,
2017
25.

Sun M., Abou-Hamad E., Rossini A.J., Zhang J., Lesage A., Zhu H., Pelletier J., Emsley L., Caps V., Basset J.
Journal of the American Chemical Society,
2013