Keywords
acyclovir
composites
release kinetics
silica
silica matrix modification
Abstract
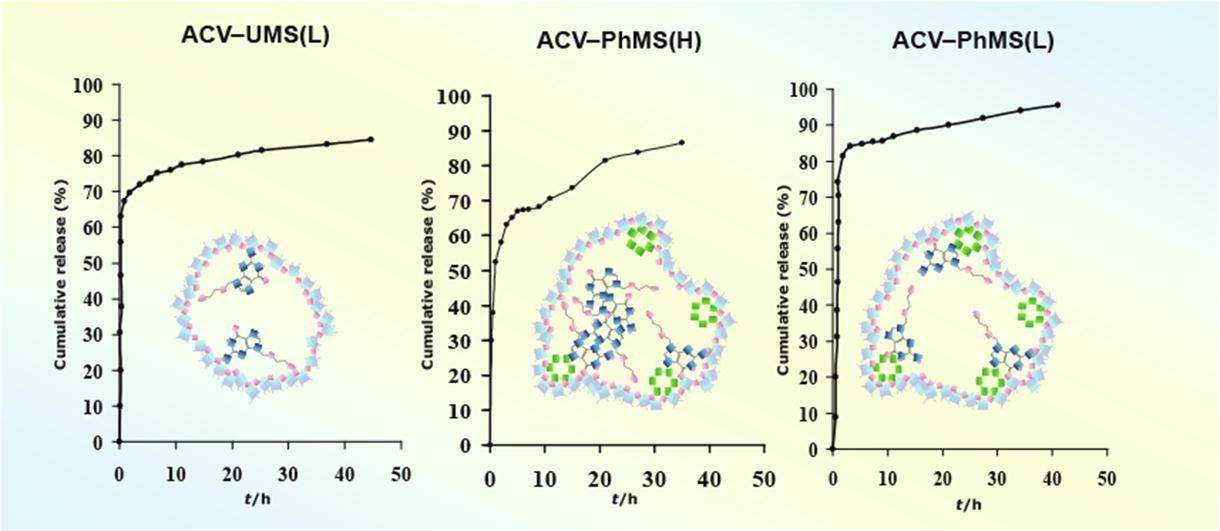
Acyclovir release from its composites with unmodified silica at pH 1.6 and 7.4 follows zero order kinetics for two days. Modification of the silica matrix with phenyl groups leads to dramatic decrease in the drug release level at pH 7.4 compared with pH 1.6 as well as to heterogeneous phase state of acyclovir in the phenyl modified composite with high loading of the drug.
References
1.

Chayavichitsilp P., Buckwalter J.V., Krakowski A.C., Friedlander S.F.
Pediatrics in Review,
2009
2.
PubChem Compound Summary. Acyclovir, https://pubchem.ncbi.nlm. nih.gov/compound/135398513.
3.

Arnal J., Gonzalez‐Alvarez I., Bermejo M., Amidon G.L., Junginger H.E., Kopp S., Midha K.K., Shah V.P., Stavchansky S., Dressman J.B., Barends D.M.
Journal of Pharmaceutical Sciences,
2008
4.
Demin A.M., Nizamov T.R., Pershina A.G., Mekhaev A.V., Uimin M.A., Minin A.S., Zakharova A.A., Krasnov V.P., Abakumov M.A., Zhukov D.G., Savchenko A.G., Schetinin I.V., Majouga A.G.
Mendeleev Communications,
2019
5.

Sábio R.M., Meneguin A.B., Ribeiro T.C., Silva R.R., Chorilli M.
International Journal of Pharmaceutics,
2019
6.
Denieva Z.G., Budanova U.A., Sebyakin Y.L.
Mendeleev Communications,
2019
7.
Strategies to Modify the Drug Release from Pharmaceutical Systems (Woodhead Publishing Series in Biomedicine, no. 85), ed. M. L. Bruschi, Woodhead Publishing, 2015, ch. 5, pp. 63–86.
8.
10.1016/j.mencom.2021.09.038_b0040
Sankar
Int. J. Pharma Bio Sci.,
2013
9.

Lembo D., Cavalli R.
Antiviral Chemistry and Chemotherapy,
2010
10.
Durai R.
Asian Journal of Pharmaceutics,
2015
11.
Food Additive Status List, U.S. Food & Drug Administration, https:// www.fda.gov/food/food-additives-petitions/food-additive-status-list#ftnC.
12.
10.1016/j.mencom.2021.09.038_b0060
Karpe
J. Appl. Pharm. Sci.,
2012
13.

Zschocke I., Reich C., Zielke A., Reitmeier N., Reich K.
Journal of Dermatological Treatment,
2008
14.
10.1016/j.mencom.2021.09.038_b0070
Bareiss
Med. J.,
2010
15.
10.1016/j.mencom.2021.09.038_b0075
Tayebee
Int. J. Nano Dimens.,
2017
16.

Lee E.C., Nguyen C.T., Strounina E., Davis-Poynter N., Ross B.P.
ACS Omega,
2018
17.
10.1016/j.mencom.2021.09.038_b0085
Dolinina
J. Pharm. Sci.,
1952
18.

Ritger P.L., Peppas N.A.
Journal of Controlled Release,
1987
19.

Zhang S., Chu Z., Yin C., Zhang C., Lin G., Li Q.
Journal of the American Chemical Society,
2013
20.

Yilmaz E., Bengisu M.
Journal of Biomedical Materials Research - Part B Applied Biomaterials,
2006
21.

Popova M.D., Szegedi Á., Kolev I.N., Mihály J., Tzankov B.S., Momekov G.T., Lambov N.G., Yoncheva K.P.
International Journal of Pharmaceutics,
2012
22.

Tang Q., Chen Y., Chen J., Li J., Xu Y., Wu D., Sun Y.
Journal of Solid State Chemistry,
2010
23.

Cauda V., Schlossbauer A., Bein T.
Microporous and Mesoporous Materials,
2010
24.

Viitala R., Jokinen M., Rosenholm J.B.
International Journal of Pharmaceutics,
2007
25.

Lutker K.M., Quiñones R., Xu J., Ramamoorthy A., Matzger A.J.
Journal of Pharmaceutical Sciences,
2011
26.
Karolewicz B., Nartowski K., Pluta J., Górniak A.
Acta Pharmaceutica,
2016
27.

Juère E., Kleitz F.
Microporous and Mesoporous Materials,
2018
28.

Maver U., Godec A., Bele M., Planinšek O., Gaberšček M., Srčič S., Jamnik J.
International Journal of Pharmaceutics,
2007
29.

Heikkilä T., Salonen J., Tuura J., Kumar N., Salmi T., Murzin D.Y., Hamdy M.S., Mul G., Laitinen L., Kaukonen A.M., Hirvonen J., Lehto V.-.
Drug Delivery,
2007