Keywords
N,N'-dimethylbarbituric acid
4-hydroxy-6-methyl-2H-pyran-2-one
benzaldehydes
morpholine
multicomponent reaction
tandem Knoevenagel–Michael reaction
Abstract
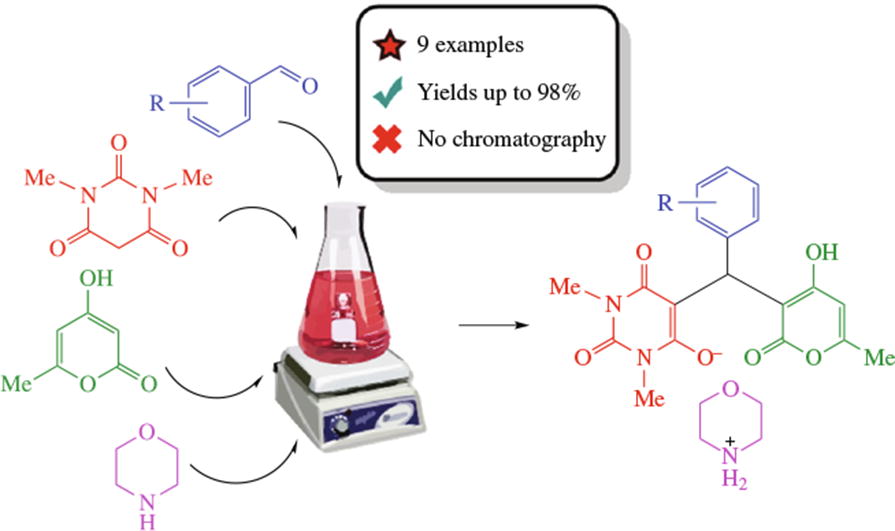
A new type of four component tandem Knoevenagel–Michael reaction has been found consisting in the assembly of benzaldehydes, N,N'-dimethylbarbituric acid, 4-hydroxy-6-methyl-2H-pyran-2-one and morpholine in alcohols, other organic solvents or water at room temperature without catalyst or any other additives, which results in the selective formation of unsymmetrical scaffold with three different heterocyclic rings in 63–98% yields. The crystal structure of morpholinium 5-[(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)(4-nitrophenyl)methyl]-1,3-dimethyl-2,6-dioxo-1,2,3,4-tetrahydropyrimidin-6-olate has been confirmed by X-ray diffraction.
References
1.
(a) Multicomponent Reactions, eds. J. Zhu and H. Bienaymé, Wiley-VCH, 2005; (b) S. Zhi, X. Ma and W. Zhang, Org. Biomol. Chem., 2019, 17, 7632; (c) V. G. Nenajdenko, Russ. Chem. Rev., 2020, 89, 1274; (d) M. N. Elinson, Yu. E. Ryzhkova and F. V. Ryzhkov, Russ. Chem. Rev., 2021, 90, 94.
2.
10.1016/j.mencom.2021.09.035_h0010
Ho
Tandem Organic Reactions,
1992
3.
Tietze L.F., Beifuss U.
1993
4.

Jadhav S.N., Patil S.P., Sahoo D.P., Rath D., Parida K., Rode C.V.
Catalysis Letters,
2020
5.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Leonova N.A., Egorov M.P.
Mendeleev Communications,
2020
6.

Liu M., Liu C., Zhang J., Xu Y., Dong L.
Organic Chemistry Frontiers,
2019
7.
10.1016/j.mencom.2021.09.035_b0030
Maleki
Appl. Organomet. Chem.,
2018
8.

Elinson M.N., Dorofeev A.S., Nasybullin R.F., Feducovich S.K., Nikishin G.I.
Electrochimica Acta,
2008
9.

Evans B.E., Rittle K.E., Bock M.G., DiPardo R.M., Freidinger R.M., Whitter W.L., Lundell G.F., Veber D.F., Anderson P.S., Chang R.S., Lotti V.J., Cerino D.J., Chen T.B., Kling P.J., Kunkel K.A., et. al.
Journal of Medicinal Chemistry,
1988
10.

Nicolaou K.C., Pfefferkorn J.A., Roecker A.J., Cao G.-., Barluenga S., Mitchell H.J.
Journal of the American Chemical Society,
2000
11.
10.1016/j.mencom.2021.09.035_b0050
Katsamakas
Med. Chem.,
2019
12.
L. L. Brunton, J. S. Lazo and K. L. Parker, Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 11th edn., McGraw-Hill Professional, 2004.
13.

14.
10.1016/j.mencom.2021.09.035_b0065
Uhlmann
Ther.,
2009
15.

Naguib F.N., Levesque D.L., Eng-Chi Wang, Panzica R.P., El Kouni M.H.
Biochemical Pharmacology,
1993
16.
10.1016/j.mencom.2021.09.035_b0075
Sriram
J. Pharm. Pharm. Sci.,
2005
17.

Pyrimidine-2,4,6-Triones: A New Effective and Selective Class of Matrix Metalloproteinase Inhibitors
Grams F., Brandstetter H., DAlò S., Geppert D., Krell H., Leinert H., Livi V., Menta E., Oliva A., Zimmermann G.
Biological Chemistry,
2001
18.
10.1016/j.mencom.2021.09.035_b0085
Liu
Cancer Res.,
1998
19.

Goekjian P.G., Jirousek M.R.
Current Medicinal Chemistry,
2022
20.

Gruber P., Rechfeld F., Kirchmair J., Hauser N., Boehler M., Garczarczyk D., Langer T., Hofmann J.
Journal of Biochemistry,
2010
21.

Vara Prasad J.V., Pavlovsky A., Para K.S., Ellsworth E.L., Tummino P.J., Nouhan C., Ferguson D.
Bioorganic and Medicinal Chemistry Letters,
1996
22.
10.1016/j.mencom.2021.09.035_b0105
Lan
Int. J. Clin. Exp. Pathol.,
2015
23.
Yin P., Liu X., Qiu Y., Cai J., Qin J., Zhu H., Li Q.
Asian Pacific Journal of Cancer Prevention,
2012
24.

TSUCHIYA K., KOBAYASHI S., NISHIKIORI T., NAKAGAWA T., TATSUTA K.
Journal of Antibiotics,
2012
25.

Kondoh M., Usui T., Kobayashi S., Tsuchiya K., Nishikawa K., Nishikiori T., Mayumi T., Osada H.
Cancer Letters,
1998
26.

Turner S.R., Strohbach J.W., Tommasi R.A., Aristoff P.A., Johnson P.D., Skulnick H.I., Dolak L.A., Seest E.P., Tomich P.K., Bohanon M.J., Horng M., Lynn J.C., Chong K., Hinshaw R.R., Watenpaugh K.D., et. al.
Journal of Medicinal Chemistry,
1998
27.

Kourounakis A.P., Xanthopoulos D., Tzara A.
Medicinal Research Reviews,
2020
28.

Cotten J.F., Keshavaprasad B., Laster M.J., Eger E.I., Yost C.S.
Anesthesia and Analgesia,
2006
29.

Stoops W.W., Strickland J.C., Alcorn J.L., Hays L.R., Rayapati A.O., Lile J.A., Rush C.R.
Psychopharmacology,
2019
30.

Bonnet U.
CNS Drug Reviews,
2003
31.

Naidu C., Kulkarni J.
Australian and New Zealand Journal of Psychiatry,
2019
32.

Walsh S.L., Heilig M., Nuzzo P.A., Henderson P., Lofwall M.R.
Addiction Biology,
2012
33.

Barakat A., Al-Majid A.M., Al-Najjar H.J., Mabkhot Y.N., Javaid S., Yousuf S., Choudhary M.I.
European Journal of Medicinal Chemistry,
2014
34.

Elinson M.N., Sokolova O.O., Nasybullin R.F.
Heterocyclic Communications,
2015
35.
Elinson M.N., Vereshchagin A.N., Ryzhkova Y.E., Krymov S.K., Leonova N.A., Goloveshkin A.S., Egorov M.P.
Mendeleev Communications,
2020
36.
(c) M. N. Elinson, R. F. Nasybullin, F. V. Ryzhkov and M. P. Egorov, C. R. Chim., 2014, 17, 437
37.
![Electrocatalytic cascade multicomponent assembling : stereoselective one-pot synthesis of the substituted 3-azabicyclo[3.1.0]hexane-1-carboxylate system from aldehyde, malononitrile, malonate and methanol](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Zaimovskaya T.A., Nikishin G.I.
Tetrahedron,
2008
38.

Elinson M.N., Nasybullin R.F., Nikishin G.I.
Comptes Rendus Chimie,
2013
39.

Elinson M.N., Merkulova V.M., Ilovaisky A.I., Barba F., Batanero B.
Electrochimica Acta,
2011
40.

Dorofeev A., Elinson M., Nasybullin R., Nikishin G.
Synthesis,
2008