Keywords
allylboration
benzylic alcohols
Friedel–Crafts reaction
lignan analogues
organoboron compounds
Abstract
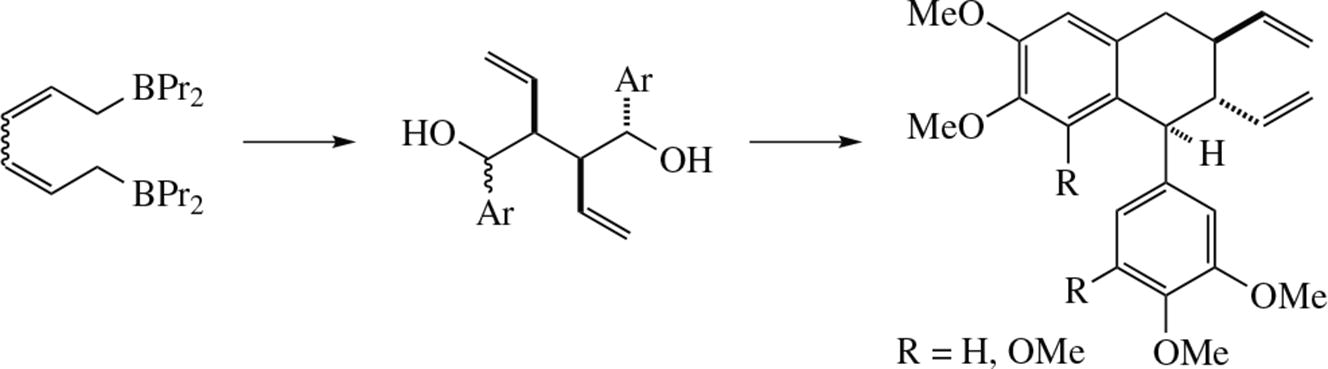
A novel convenient two-stage synthesis of aryltetraline lignin analogues from aromatic aldehydes and diallyl diboron derivatives on the basis of intramolecular Friedel–Crafts reaction has been developed.
References
1.
10.1016/j.mencom.2021.09.033_b0005
Mikhailov
Bororganicheskie soedineniya v orgsnicheskom sinteze (Organoboron Compounds in Organic Synthesis),
1977
2.
10.1016/j.mencom.2021.09.033_b0010
Mikhailov
Organoboron Compounds in Organic Synthesis,
1984
3.
10.1016/j.mencom.2021.09.033_b0015
Science of Synthesis,
2004
4.
10.1016/j.mencom.2021.09.033_b0020
Pelter
Borane Reagents,
1988
5.
10.1016/j.mencom.2021.09.033_b0025
Matteson
Stereodirected Synthesis with Organoboranes,
1995
6.

Bubnov Y.N.
Pure and Applied Chemistry,
1987
7.

8.
10.1016/j.mencom.2021.09.033_b0040
Comprehensive Heterocyclic Chemistry-II,
1996
9.

Brown H.C., Jadhav P.K.
Journal of the American Chemical Society,
1983
10.

Racherla U.S., Brown H.C.
Journal of Organic Chemistry,
1991
11.

Brown H.C., Ramachandran P.V.
Pure and Applied Chemistry,
1994
12.
10.1016/j.mencom.2021.09.033_b0060
Comprehensive Organic Synthesis,
1991
13.
Yu. N. Bubnov, M. E. Gurskii and A. V. Geiderikh, Organomet. Chem. USSR, 1989, 2, 761 (Metalloorg. Khim., 1989, 2, 1433).
14.

Gursky M.E., Geiderikh A.V., Ignatenko A.V., Bubnov Y.N.
Russian Chemical Bulletin,
1993
15.
M. E. Gursky, A. V. Geiderikh, S. B. Golovin, A. V. Ignatenko and Yu. N. Bubnov, Russ. Chem. Bull., 1993, 42, 215 (Izv. Akad. Nauk, Ser. Khim., 1993, 236).
16.
Anfimov A.N., Erdyakov S.Y., Gurskii M.E., Ignatenko A.V., Lyssenko K.A., Bubnov Y.N.
Mendeleev Communications,
2011
17.
A. N. Anfimov, S. Yu. Erdyakov, M. E. Gurskii and Yu. N. Bubnov, Russ. Chem. Bull., Int. Ed., 2011, 60, 2336 (Izv. Akad. Nauk, Ser. Khim., 2011, 2291).
18.

Hearon W.M., MacGregor W.S.
Chemical Reviews,
1955
19.

Ward R.S.
Chemical Society Reviews,
1982
20.

Ward R.S.
Natural Product Reports,
1993
21.

Ward R.S.
Natural Product Reports,
1999
22.
10.1016/j.mencom.2021.09.033_b0110
Ayres
Lignans: Chemical,
1990
23.

Pan J., Chen S., Yang M., Wu J., Sinkkonen J., Zou K.
Natural Product Reports,
2009
24.

Saleem M., Kim H.J., Ali M.S., Lee Y.S.
Natural Product Reports,
2005
25.

26.

Cheshkov D.A., Sheberstov K.F., Sinitsyn D.O., Chertkov V.A.
Magnetic Resonance in Chemistry,
2018
27.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Jr. Montgomery, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.02, Gaussian, Wallingford, CT, 2009.
28.

Soorukram D., Pohmakotr M., Kuhakarn C., Reutrakul V.
Synthesis,
2018
29.

Biftu T., Hazra B.G., Stevenson R., Williams J.R.
Journal of the Chemical Society Perkin Transactions 1,
1978
30.

Perry C.W., Kalnins M.V., Deitcher K.H.
Journal of Organic Chemistry,
1972
31.

Jagtap P.R., Císařová I., Jahn U.
Organic and Biomolecular Chemistry,
2018
32.

Hetzler B.E., Volpin G., Vignoni E., Petrovic A.G., Proni G., Hu C.T., Trauner D.
Angewandte Chemie - International Edition,
2018
33.

Sellars J.D., Steel P.G.
European Journal of Organic Chemistry,
2007
34.

Liu X., Chen P., Li X., Ba M., Jiao X., Guo Y., Xie P.
Bioorganic and Medicinal Chemistry Letters,
2018
35.

Li X., Jiao X., Liu X., Tian C., Dong L., Yao Y., Xie P.
Tetrahedron Letters,
2014
36.

Hong B., Hsu C., Lee G.
Chemical Communications,
2012
37.

Coltart D.M., Charlton J.L.
Canadian Journal of Chemistry,
1996
38.

Rye C.E., Barker D.
Journal of Organic Chemistry,
2011