Keywords
Michael addition
nitro compounds
nitrosoalkenes
unsaturated oximes
ylides
Abstract
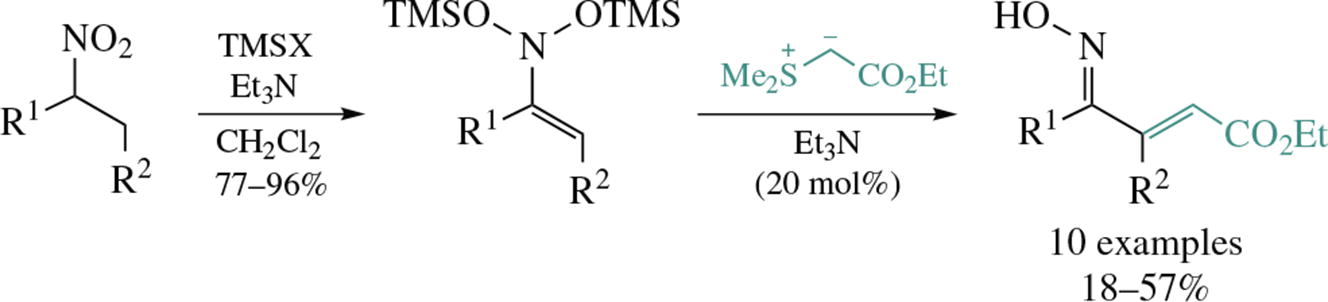
A novel approach to conjugated enone oximes from aliphatic nitro compounds deals with double silylation of N,N-bis(silyloxy) enamines followed by a stereoselective reaction with an ester-stabilized sulfur ylide. The proposed mechanism involves the generation of labile nitrosoalkenes as intermediates, which react with the sulfur ylide to give target enone oximes.
References
1.

Parthasarathy K., Jeganmohan M., Cheng C.
Organic Letters,
2007
2.

Martin R.M., Bergman R.G., Ellman J.A.
Journal of Organic Chemistry,
2012
3.

Shi Z., Koester D.C., Boultadakis-Arapinis M., Glorius F.
Journal of the American Chemical Society,
2013
4.

Liu S., Liebeskind L.S.
Journal of the American Chemical Society,
2008
5.

Yoshida Y., Kurahashi T., Matsubara S.
Chemistry Letters,
2012
6.

Neely J.M., Rovis T.
Journal of the American Chemical Society,
2012
7.

Neely J.M., Rovis T.
Journal of the American Chemical Society,
2014
8.

Zhang L., Duan J., Xu G., Ding X., Mao Y., Rong B., Zhu N., Fang Z., Li Z., Guo K.
Journal of Organic Chemistry,
2020
9.
![NH4I-Triggered [4 + 2] Annulation of α,β-Unsaturated Ketoxime Acetates with N-Acetyl Enamides for the Synthesis of Pyridines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Duan J., Zhang L., Xu G., Chen H., Ding X., Mao Y., Rong B., Zhu N., Guo K.
Journal of Organic Chemistry,
2020
10.

Golovanov A.A., Odin I.S., Zlotskii S.S.
Russian Chemical Reviews,
2019
11.

Danilenko V.M., Tishkov A.A., Ioffe S.L., Lyapkalo I.M., Strelenko Y.A., Tartakovsky V.A.
Synthesis,
2002
12.

Lerche H., Treiber J., Severin T.
Chemische Berichte,
1980
13.

Sosnovskikh V.Y., Usachev B.I., Sizov A.Y., Kodess M.I.
Tetrahedron Letters,
2004
14.

Bravo P., Gaudiano G., Ponti P.P., Ticozzi C.
Tetrahedron,
1972
15.
10.1016/j.mencom.2021.09.031_b0075
Sukhorukov
Synlett,
2020
16.

Zlotin S.G., Dalinger I.L., Makhova N.N., Tartakovsky V.A.
Russian Chemical Reviews,
2020
17.
![Synthesis of Isoxazolines from Nitroalkanes via a [4+1]‐Annulation Strategy](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ushakov P.Y., Khatuntseva E.A., Nelyubina Y.V., Tabolin A.A., Ioffe S.L., Sukhorukov A.Y.
Advanced Synthesis and Catalysis,
2019
18.

Buono F.G., Eriksson M.C., Yang B., Kapadia S.R., Lee H., Brazzillo J., Lorenz J.C., Nummy L., Busacca C.A., Yee N., Senanayake C.
Organic Process Research and Development,
2014
19.

Dilman A.D., Tishkov A.A., Lyapkalo I.M., Ioffe S.L., Strelenko Y.A., Tartakovsky V.A.
Synthesis,
1998
20.

Dugave C., Demange L.
Chemical Reviews,
2003
21.

Calò V., Nacci A., Monopoli A., Lopez L., di Cosmo A.
Tetrahedron,
2001
22.

Lopes S.M., Cardoso A.L., Lemos A., Pinho e Melo T.M.
Chemical Reviews,
2018
23.

Boyko Y.D., Dorokhov V.S., Sukhorukov A.Y., Ioffe S.L.
Beilstein Journal of Organic Chemistry,
2017
24.

Lesiv A., Ioffe S., Strelenko Y., Tartakovsky V.
Helvetica Chimica Acta,
2002
25.

Zhmurov P.A., Khoroshutina Y.A., Novikov R.A., Golovanov I.S., Sukhorukov A.Y., Ioffe S.L.
Chemistry - A European Journal,
2017
26.

Bhujle V., Wild U.P., Baumann H., Wagnière G.
Tetrahedron,
1976
27.

Nunes S.C., Lopes S.M., Gomes C.S., Lemos A., Pais A.A., Pinho e Melo T.M.
Journal of Organic Chemistry,
2014