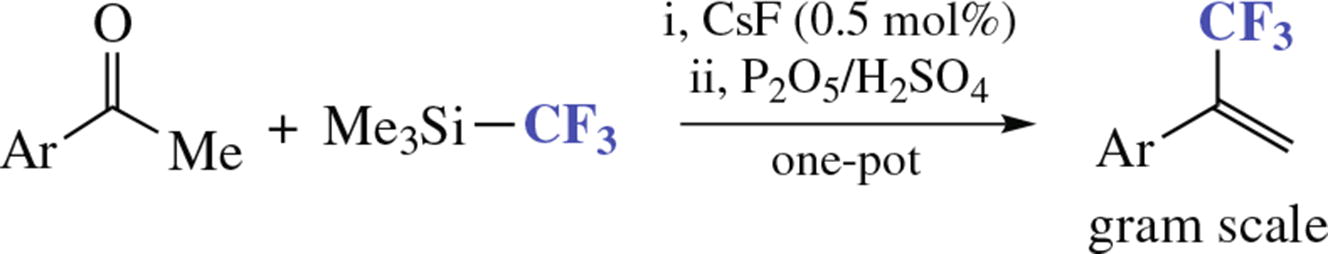Keywords
alkenes
Ketones
organofluorine compounds
silicon reagents
trifluoromethylation
Abstract
A new synthesis of α-trifluoromethylstyrenes from aromatic ketones and (trifluoromethyl) trimethylsilane is described. The reaction involves nucleophilic trifluoromethylation and elimination of trimethylsilanol, which are performed within one reaction flask.
References
1.

Inoue M., Sumii Y., Shibata N.
ACS Omega,
2020
2.

Ogawa Y., Tokunaga E., Kobayashi O., Hirai K., Shibata N.
iScience,
2020
3.

Han J., Remete A.M., Dobson L.S., Kiss L., Izawa K., Moriwaki H., Soloshonok V.A., O’Hagan D.
Journal of Fluorine Chemistry,
2020
4.

Politanskaya L.V., Selivanova G.A., Panteleeva E.V., Tretyakov E.V., Platonov V.E., Nikul’shin P.V., Vinogradov A.S., Zonov Y.V., Karpov V.M., Mezhenkova T.V., Vasilyev A.V., Koldobskii A.B., Shilova O.S., Morozova S.M., Burgart Y.V., et. al.
Russian Chemical Reviews,
2019
5.

Tian F., Yan G., Yu J.
Chemical Communications,
2019
6.
10.1016/j.mencom.2021.09.030_h0015
Bégué
Tetrahedron Lett.,
1995
7.

Fuchibe K., Takahashi M., Ichikawa J.
Angewandte Chemie - International Edition,
2012
8.

Chen Y., Ni N., Cheng D., Xu X.
Tetrahedron Letters,
2020
9.

Wiles R.J., Phelan J.P., Molander G.A.
Chemical Communications,
2019
10.

Lin Z., Lan Y., Wang C.
Organic Letters,
2020
11.

Supranovich V.I., Levin V.V., Kokorekin V.A., Dilman A.D.
Advanced Synthesis and Catalysis,
2021
12.

Miura T., Ito Y., Murakami M.
Chemistry Letters,
2008
13.

Lan Y., Yang F., Wang C.
ACS Catalysis,
2018
14.

Rabasa-Alcañiz F., Asensio A., Sánchez-Roselló M., Escolano M., del Pozo C., Fustero S.
Journal of Organic Chemistry,
2017
15.

Cyr P., Flynn-Robitaille J., Boissarie P., Marinier A.
Organic Letters,
2019
16.

Paratian J.M., Labbé E., Sibille S., Périchon J.
Journal of Organometallic Chemistry,
1995
17.
10.1016/j.mencom.2021.09.030_h0055
Lishchynskyi
Synlett,
2015
18.

Jiang B., Xu Y.
Journal of Organic Chemistry,
1991
19.

Pan R., Liu X., Deng M.
Journal of Fluorine Chemistry,
1999
20.

Kobayashi O., Uraguchi D., Yamakawa T.
Journal of Fluorine Chemistry,
2009
21.

Wang X., Xu Y., Deng Y., Zhou Y., Feng J., Ji G., Zhang Y., Wang J.
Chemistry - A European Journal,
2013
22.

Nader B.S., Cordova J.A., Reese K.E., Powell C.L.
Journal of Organic Chemistry,
1994
23.

TARRANT P., TAYLOR R.E.
Journal of Organic Chemistry,
1959
24.

Kazennikova G.V., Talalaeva T.V., Zimin A.V., Kocheshkov K.A.
Russian Chemical Bulletin,
1961
25.

Sigan A.L., Golubev A.S., Belyaeva E.V., Gorfinkel S.M., Kagramanov N.D., Spiridonov Y.Y., Chkanikov N.D.
Russian Chemical Bulletin,
2019
26.

NAGAI T., HAMA M., YOSHIOKA M., YUDA M., YOSHIDA N., ANDO A., KOYAMA M., MIKI T., KUMADAKI I.
Chemical and Pharmaceutical Bulletin,
2011
27.

Prakash G.K., Yudin A.K.
Chemical Reviews,
1997
28.

Liu X., Xu C., Wang M., Liu Q.
Chemical Reviews,
2014
29.

Singh R.P., Shreeve J.M.
Tetrahedron,
2000
30.

Dilman A.D., Levin V.V.
European Journal of Organic Chemistry,
2011
31.

Kolomeitsev A., Movchun V., Rusanov E., Bissky G., Lork E., Röschenthaler G., Kirsch P.
Chemical Communications,
1999
32.
![[Me3Si(CF3)F]− and [Me3Si(CF3)2]−: Reactive Intermediates in Fluoride-Initiated Trifluoromethylation with Me3SiCF3— An NMR Study](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Maggiarosa N., Tyrra W., Naumann D., Kirij N.V., Yagupolskii Y.L.
Angewandte Chemie - International Edition,
1999
33.

Prakash G.K., Wang F., Zhang Z., Haiges R., Rahm M., Christe K.O., Mathew T., Olah G.A.
Angewandte Chemie - International Edition,
2014
34.

Prakash G.K., Krishnamurti R., Olah G.A.
Journal of the American Chemical Society,
1989
35.

Prakash G.K., Panja C., Vaghoo H., Surampudi V., Kultyshev R., Mandal M., Rasul G., Mathew T., Olah G.A.
Journal of Organic Chemistry,
2006