Keywords
1,2,3-dithiazoles
bond orders
disulfide bond
electron delocalization indices
turning radii
Abstract
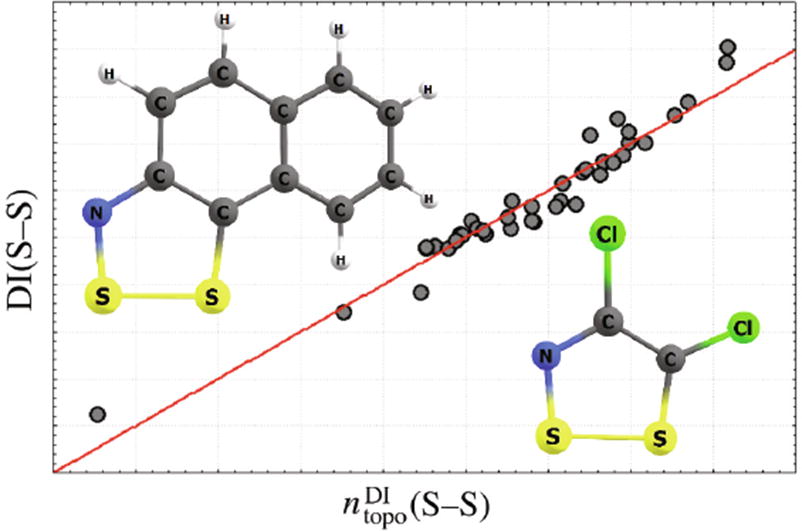
A parametric QTAIM-based (topological) model of bond orders and a modification of the Pauling bond order model are proposed for N,S-containing heterocycles, in particular, for 1,2,3-dithiazoles and 1,2,3-dithiazolium systems, which are prone to the formation of stable radicals and therefore are promising compounds in photovoltaics. These models have been parameterized for covalent S–N, S–C and S–S bonds using the electron delocalization indices. A modified Pauling’s bond order model uses turning radii, that is, the distances within which the potential acting on an electron in a system still tends to return that electron to the atomic basin, and avoids the need to choose the hybridization state of bound atoms arbitrarily.
References
1.

Konstantinova L.S., Rakitin O.A.
Russian Chemical Reviews,
2008
2.

Konstantinova L., Baranovsky I., Irtegova I., Bagryanskaya I., Shundrin L., Zibarev A., Rakitin O.
Molecules,
2016
3.

Barclay T.M., Cordes A.W., Goddard J.D., Mawhinney R.C., Oakley R.T., Preuss K.E., Reed R.W.
Journal of the American Chemical Society,
1997
4.

Mailman A., Robertson C.M., Winter S.M., Dube P.A., Oakley R.T.
Inorganic Chemistry,
2019
5.
Volkova Y.M., Makarov A.Y., Pritchina E.A., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2020
6.

Knyazeva E.A., Rakitin O.A.
Russian Chemical Reviews,
2016
7.

Barclay T.M., Wallace Cordes A., Beer L., Oakley R.T., Preuss K.E., Taylor N.J., Reed R.W.
Chemical Communications,
1999
8.
10.1016/j.mencom.2021.09.029_b0040
Stable Radicals: Fundamentals and Applied Aspects of Odd-Electron Compounds,
2010
9.

Rakitin O.A.
Russian Chemical Reviews,
2011
10.

Makarov A.Y., Chulanova E.A., Semenov N.A., Pushkarevsky N.A., Lonchakov A.V., Bogomyakov A.S., Irtegova I.G., Vasilieva N.V., Lork E., Gritsan N.P., Konchenko S.N., Ovcharenko V.I., Zibarev A.V.
Polyhedron,
2014
11.
Novel long-lived π-heterocyclic radical anion: a hybrid of 1,2,5-thiadiazo- and 1,2,3-dithiazolidyls
Chulanova E.A., Irtegova I.G., Vasilieva N.V., Bagryanskaya I.Y., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2015
12.

Dawood K.M., Farghaly T.A.
Expert Opinion on Therapeutic Patents,
2017
13.

Jain A.K., Sharma S., Vaidya A., Ravichandran V., Agrawal R.K.
Chemical Biology and Drug Design,
2013
14.

Heering W., Usleber E., Dietrich R., Märtlbauer E.
The Analyst,
1998
15.

Gleiter R., Haberhauer G.
Journal of Organic Chemistry,
2014
16.

Shukla R., Dhaka A., Aubert E., Vijayakumar-Syamala V., Jeannin O., Fourmigué M., Espinosa E.
Crystal Growth and Design,
2020
17.

Gleiter R., Haberhauer G.
Coordination Chemistry Reviews,
2017
18.

Pauling L.
Journal of the American Chemical Society,
1947
19.

Ospadov E., Tao J., Staroverov V.N., Perdew J.P.
Proceedings of the National Academy of Sciences of the United States of America,
2018
20.

Zhao D., Yang Z.
Journal of Computational Chemistry,
2014
21.

Zhao D., Yan C., Zhu Z., Zhang L., Jiang Y., Gong R., Yang Z.
Molecular Physics,
2018
22.

Zhao D., Gong L., Yang Z.
Journal of Physical Chemistry A,
2005
23.

24.

Mayer I.
Chemical Physics Letters,
2012
25.

Giambiagi M., de Giambiagi M.S., Mundim K.C.
Structural Chemistry,
1990
26.

Bartashevich E.V., Troitskaya E.A., Tsirelson V.G.
Chemical Physics Letters,
2014
27.

Cioslowski J., Mixon S.T.
Journal of the American Chemical Society,
1991
28.

Matta C.F., Hernández-Trujillo J.
Journal of Physical Chemistry A,
2003
29.

Matta C.F., Hernández-Trujillo J.
Journal of Physical Chemistry A,
2005
30.

Bader R.F., Slee T.S., Cremer D., Kraka E.
Journal of the American Chemical Society,
1983
31.

Matta C.F.
Journal of Computational Chemistry,
2014
32.

Howard S.T., Lamarche O.
Journal of Physical Organic Chemistry,
2003
33.

Fradera X., Austen M.A., Bader R.F.
Journal of Physical Chemistry A,
1998
34.

35.
10.1016/j.mencom.2021.09.029_b0175
The Quantum Theory of Atoms in Molecules
From Solid State to DNA and Drug Design, eds,
2007
36.

Tsirelson V.G., Stash A.I., Potemkin V.A., Rykounov A.A., Shutalev A.D., Zhurova E.A., Zhurov V.V., Pinkerton A.A., Gurskaya G.V., Zavodnik V.E.
Acta Crystallographica Section B Structural Science,
2006
37.

Tsirelson V.G., Bartashevich E.V., Stash A.I., Potemkin V.A.
Acta Crystallographica Section B Structural Science,
2007
38.

Stash A., Tsirelson V.
Journal of Applied Crystallography,
2002
39.

Stash A.I., Tsirelson V.G.
Journal of Applied Crystallography,
2014
40.

Zhurova E.A., Stash A.I., Tsirelson V.G., Zhurov V.V., Bartashevich E.V., Potemkin V.A., Pinkerton A.A.
Journal of the American Chemical Society,
2006
41.

Milašinović V., Krawczuk A., Molčanov K., Kojić-Prodić B.
Crystal Growth and Design,
2020
42.

Mukhitdinova S.E., Bartashevich E.V., Tsirelson V.G.
Journal of Structural Chemistry,
2018
43.

Bartashevich E.V., Nikulov D.K., Vener M.V., Tsirelson V.G.
Computational and Theoretical Chemistry,
2011
44.

Bader R.F., Stephens M.E.
Journal of the American Chemical Society,
1975
45.

Cordero B., Gómez V., Platero-Prats A.E., Revés M., Echeverría J., Cremades E., Barragán F., Alvarez S.
Dalton Transactions,
2008
46.
A. A. Granovsky, Firefly computational chemistry program, version 8.0.1, 2014. http://classic.chem.msu.su/gran/gamess/index.html.
47.
T. A. Keith, AIMAll Professional quantum chemistry software package, TK Gristmill Software, version 15.09.27, 2015. http://aim. tkgristmill.com/index.html.
48.
STATISTICA: Data Mining, StatSoft Russia. http://statsoft.ru/.