Keywords
calculations
DFT
hydrazones
hydrogen bonding
keto esters
methanolysis
QTAIM
Abstract
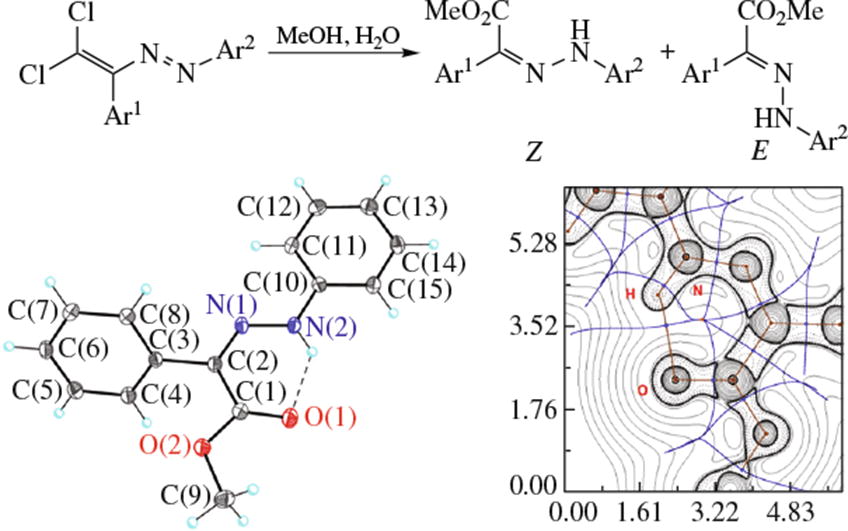
Novel approach to methyl 2-aryl-2-(arylhydrazono) acetates via methanolysis of dichlorodiazabutadienes was developed. Hydrogen bonding in methyl Z-2-phenyl-2-(phenyl-hydrazono) acetate was explored by DFT calculations and topological analysis of the electron density distribution within the framework of Bader’s theory (QTAIM method).
References
1.

Enders D., Wortmann L., Peters R.
Accounts of Chemical Research,
2000
2.

Lazny R., Nodzewska A.
Chemical Reviews,
2009
3.

Kobayashi S., Mori Y., Fossey J.S., Salter M.M.
Chemical Reviews,
2011
4.

Vicini P., Zani F., Cozzini P., Doytchinova I.
European Journal of Medicinal Chemistry,
2002
5.

Savini L., Chiasserini L., Travagli V., Pellerano C., Novellino E., Cosentino S., Pisano M.B.
European Journal of Medicinal Chemistry,
2004
6.

Tskhovrebov A.G., Novikov A.S., Odintsova O.V., Mikhaylov V.N., Sorokoumov V.N., Serebryanskaya T.V., Starova G.L.
Journal of Organometallic Chemistry,
2019
7.

Lehn J.
Angewandte Chemie - International Edition,
2013
8.

Repina O.V., Novikov A.S., Khoroshilova O.V., Kritchenkov A.S., Vasin A.A., Tskhovrebov A.G.
Inorganica Chimica Acta,
2020
9.

Zhou X., Wu Y., Li D.
Journal of the American Chemical Society,
2013
10.

Lygaitis R., Getautis V., Grazulevicius J.V.
Chemical Society Reviews,
2008
11.

Hughes D.L.
Organic Preparations and Procedures International,
1993
12.

Japp F.R., Klingemann F.
Berichte der deutschen chemischen Gesellschaft,
1887
13.

Wagaw S., Yang B.H., Buchwald S.L.
Journal of the American Chemical Society,
1998
14.

Nenajdenko V.G., Shastin A.V., Gorbachev V.M., Shorunov S.V., Muzalevskiy V.M., Lukianova A.I., Dorovatovskii P.V., Khrustalev V.N.
ACS Catalysis,
2016
15.

Sergeev P.G., Khrustalev V.N., Nenajdenko V.G.
European Journal of Organic Chemistry,
2020
16.

Shikhaliyev N.Q., Ahmadova N.E., Gurbanov A.V., Maharramov A.M., Mammadova G.Z., Nenajdenko V.G., Zubkov F.I., Mahmudov K.T., Pombeiro A.J.
Dyes and Pigments,
2018
17.

Shastin A.V., Tsyrenova B.D., Sergeev P.G., Roznyatovsky V.A., Smolyar I.V., Khrustalev V.N., Nenajdenko V.G.
Organic Letters,
2018
18.

Shikhaliyev N.Q., Kuznetsov M.L., Maharramov A.M., Gurbanov A.V., Ahmadova N.E., Nenajdenko V.G., Mahmudov K.T., Pombeiro A.J.
CrystEngComm,
2019
19.

Shastin A.V., Sergeev P.G., Lukianova A.I., Muzalevskiy V.M., Khrustalev V.N., Dorovatovskii P.V., Nenajdenko V.G.
European Journal of Organic Chemistry,
2018
20.
Tsyrenova B.D., Tarasevich B.N., Khrustalev V.N., Gloriozov I.P., Nenajdenko V.G.
Mendeleev Communications,
2020
21.

Nenajdenko V.G., Shikhaliyev N.G., Maharramov A.M., Bagirova K.N., Suleymanova G.T., Novikov A.S., Khrustalev V.N., Tskhovrebov A.G.
Molecules,
2020
22.
Shikhaliyev N.G., Maharramov A.M., Bagirova K.N., Suleymanova G.T., Tsyrenova B.D., Nenajdenko V.G., Novikov A.S., Khrustalev V.N., Tskhovrebov A.G.
Mendeleev Communications,
2021
23.

Maharramov A.M., Shikhaliyev N.Q., Suleymanova G.T., Gurbanov A.V., Babayeva G.V., Mammadova G.Z., Zubkov F.I., Nenajdenko V.G., Mahmudov K.T., Pombeiro A.J.
Dyes and Pigments,
2018
24.
10.1016/j.mencom.2021.09.028_b0120
Anh
Acta Crystallogr.,
2012
25.
10.1016/j.mencom.2021.09.028_b0125
Vickery
J. Chem. Soc., Perkin Trans.,
1454
26.

Mikhaylov V.N., Sorokoumov V.N., Novikov A.S., Melnik M.V., Tskhovrebov A.G., Balova I.A.
Journal of Organometallic Chemistry,
2020
27.

Luzyanin K.V., Tskhovrebov A.G., Carias M.C., Guedes da Silva M.F., Pombeiro A.J., Kukushkin V.Y.
Organometallics,
2009
28.

Tskhovrebov A.G., Novikov A.S., Tupertsev B.S., Nazarov A.A., Antonets A.A., Astafiev A.A., Kritchenkov A.S., Kubasov A.S., Nenajdenko V.G., Khrustalev V.N.
Inorganica Chimica Acta,
2021
29.

Astafiev A.A., Repina O.V., Tupertsev B.S., Nazarov A.A., Gonchar M.R., Vologzhanina A.V., Nenajdenko V.G., Kritchenkov A.S., Khrustalev V.N., Nadtochenko V.N., Tskhovrebov A.G.
Molecules,
2021
30.

Tskhovrebov A.G., Goddard R., Fürstner A.
Angewandte Chemie - International Edition,
2018
31.

Tskhovrebov A.G., Solari E., Scopelliti R., Severin K.
Organometallics,
2014
32.

Liu Y., Varava P., Fabrizio A., Eymann L.Y., Tskhovrebov A.G., Planes O.M., Solari E., Fadaei-Tirani F., Scopelliti R., Sienkiewicz A., Corminboeuf C., Severin K.
Chemical Science,
2019
33.
10.1016/j.mencom.2021.09.028_b0165
Serebryanskaya
Mater.,
2020
34.

Mikhaylov V., Sorokoumov V., Liakhov D., Tskhovrebov A., Balova I.
Catalysts,
2018
35.

Tskhovrebov A.G., Vasileva A.A., Goddard R., Riedel T., Dyson P.J., Mikhaylov V.N., Serebryanskaya T.V., Sorokoumov V.N., Haukka M.
Inorganic Chemistry,
2018
36.

Brandenburg J.G., Bannwarth C., Hansen A., Grimme S.
Journal of Chemical Physics,
2018
37.

Bader R.F.
Chemical Reviews,
1991
38.

Johnson E.R., Keinan S., Mori-Sánchez P., Contreras-García J., Cohen A.J., Yang W.
Journal of the American Chemical Society,
2010
39.

Espinosa E., Molins E., Lecomte C.
Chemical Physics Letters,
1998
40.

Steiner T.
Angewandte Chemie - International Edition,
2002
41.
10.1016/j.mencom.2021.09.028_b0205
Temesgen
Acta Crystallogr.,
2021
42.

Tskhovrebov A.G., Novikov A.S., Khrustalev V.N.
Journal of Structural Chemistry,
2021
43.

Espinosa E., Alkorta I., Elguero J., Molins E.
Journal of Chemical Physics,
2002
44.

Contreras-García J., Johnson E.R., Keinan S., Chaudret R., Piquemal J., Beratan D.N., Yang W.
Journal of Chemical Theory and Computation,
2011
45.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
46.
Bruker SAINT, Bruker AXS Inc., Madison, WI, 2018.
47.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015